2016 CHEMISTRY SUMMER SCHOOL
HILLSIDE H.S. & L.C.
Monday July 11
AGENDA:
- TEST TOMORROW ON CHAPTERS 11 AND 12 AND ALSO RESPONSIBLE FOR 1 - 22 OF STANDARDIZED TEST GIVEN BACK TO YOU
- REVIEWED ACT STANDARDIZED TEST UP TO AROUND #22
- PROBLEMS ASSIGNED SPECIFICALLY
- THE ANSWERS FOR YOUR PRE-TEST IS UNDERNEATH. JUST "CLICK" ON FILE AND RECEIVE THE WHOLE DIGITAL TEST AS WELL AS THE ANSWERS.
| chemistry_entire_act_practice_240_questions.pdf | |
| File Size: | 1736 kb |
| File Type: | |
Friday July 8
AGENDA:
Thursday July 7
AGENDA:
Wednesday July 6
AGENDA:
- Go over the written tests
- Give workbook packets out for next chapter(s)
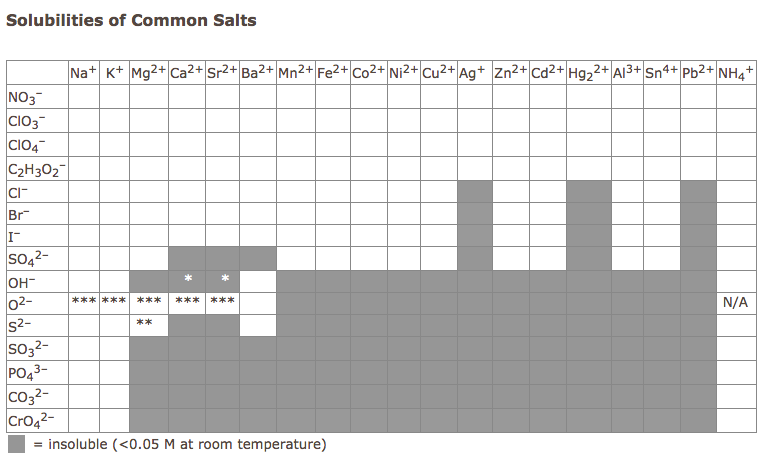
- Make and distribute solution solubility table.
- Go over pre-class standardized test questions.
- Double replacement reactions. (The cations switch their anion partners with each other)
- Metal1/Nonmetal1(aq) + Metal2/Nonmetal2(aq) ► Metal1/Nonmetal2 (unknown) + Metal2/Nonmetal1 (unknown)
- If something is soluble then it will dissolve in solution and it is aqueous (aq)
- If something is insoluble then it will not dissolve and precipitate (ppt) out of solution to become a solid (s)
- The way to find out if a compound (in these cases salt) is soluble or insoluble you must learn or look at a solubility table
- When a solid does come up as a product of the two aqueous solutions then this is also called a precipitation reaction.
Tuesday July 5
AGENDA:
- 2 PART TEST. FIRST IS 20 KAHOOT PROBLEMS. THEN 4 PROBLEMS (1 IS EXTRA CREDIT) WRITTEN. WORTH 50/50
- All grades will be finalized by tomorrow concerning your present grades including the test
- Go and finish the rest of chapter 11. Pick 2 section review problems for each section after 11.1 (we covered that already)
Friday July 1
AGENDA:
- Will be on test: A lewis diagram/VESPR question, know bond strengths even which intermolecular forces (Vanderwaals) are strongest and weakest,
- The SI (or Système International d'Unités ) is the modern form of the metric system.The metric system was originally established after the French Revolution (1789-1799) as a consistent set of decimal units with base units that could be precisely determined by reference to standards (a platinum metre stick and a platinum kilogram mass kept in Sèvres, France). The SI (officially established in 1960) differs from older versions of the metric system in the number of base units and in the way these base units are defined. For example, the older CGS system was based on centimeters, grams, and seconds; the modern SI is based on meters, kilograms, seconds, kelvins, moles, amperes, and candelas
- q=nC△T q=heat (measured in energy units, the joule); n= # of moles; C= molar heat capacity; △T= change in temperature
- △H=C△T
- △G = △H - T△S ( if change in Gibbs energy is negative it is spontaneous. When △G=0 there is equilibrium)
- Note that molar heat capacities (C) are always tabulated in units that include joules, whereas enthalpies (H) are usually reported in terms of kilojoules
- Standard conditions for thermodynamic properties are 25℃ and 100 kPa pressure (different than 1 atm)
- Melting point temperature = △Hfus/△Sfus and the boiling point temperature is △Hvap/△Svap
- The symbol for molar enthalpy is H because enthalpy was once called "heat content"
- Photosynthesis: 2870 Kj/mol + 6CO₂(g) + 6H₂O(l) ► C₆H₁₂O₆(s) + 6O₂(g); △H= 2870 kj; △Sº=-259 J/K; △Gº= 2947 kJ
- HAND WARMER- (If material available): Iron in powdered form, place 25g of it in a small sealable plastic bag, add 1g of NaCl, seal the bag and shake to mix. Add 1 tablespoon of expanded vermiculite, reseal the bag, and shake it again. To activate the hand warmer, add 5 mL of water to the bag. Seal the bag, and squeeze or shake it thoroughly to mix the contents. The heat given off in the reaction will produce a noticeable temperature increase in about 1 minute.
- QUICK LAB: MOLAR HEAT CAPACITY OF METALS...
- Choose 3 samples of metals (same from density lab). If possible, use uniform 2.5cm blocks. Measure mass of each sample, and write mass on board. Have students determine the amount in moles of each metal present as I continue the setup.
- Pour equal volumes of room temperature water into each of three 250mL beakers. use enough water to cover the samples by at least 1cm. Add thermometer to each beaker.
- Use tongs to remove samples from beaker and into a pan of boiling water on hot plate. after the water returns to a boil, allow the samples to remain in the water for 1 minute.
- Using tongs, remove each metal and return them to their beakers
- Have student watch thermometers as they swirl the beakers very gently, and record the highest temperature reached by the water in each beaker.
- Have students use their results to order the metals from highest to lowest molar heat capacity. What were their findings?
- _____ depend on how much of the material you are dealing with so the quantity must be specified, as with mass and volume, while the _____ are specific to the particular substance and do not depend on how much material is present. Density would be an example of the later
- Change in enthalpy is represented by the symbol _____
- Change in entropy is represented by the symbol _____
- For a cooling process the change in enthalpy will be a _____ number
- For an endothermic process the change in enthalpy will be a _____ number
- The unit used to express enthalpy is _______
- The heat of fusion (△Hfus) occurs during _____. It is the heat absorbed when 1 mol of a substance melts.
- The heat of vaporization (△Hvap) occurs during _____. It is the heat absorbed with 1 mol of a substance vaporizes.
- Significantly less energy is required to melt a collection of molecules than the energy required to _____ the same set of molecules.
- The _____ of a liquid is affected greatly by atmospheric pressure, whereas the change in freezing point is very small
- The melting point occurs at the temperature when △Hfus = _____
- A reaction that is not spontaneous will not occur, and a reaction that is spontaneous _____ occur
- Once green plants manufacture glucose and starch, they carry on _____ just as animals do.
- _____ is an endothermic reaction that takes place in the chloroplast._____ is an exothermic reaction that takes place in mitochondria
- The most efficient storage medium that you have when it comes to food is _____. It contains a higher energy content per gram than either carbohydrates or proteins.
- The unit that see listed under the "Nutrition facts:" label on foodstuffs are the _____ which is equivalent to 1 kcal in SI which equals 4.184 kJ.
|
fats
may boiling extensive properties negative |
Calorie
cellular respiration vaporize intensive properties positive |
photosynthesis
boiling point '△H kilojoules |
cellular respiration
T△Sfus △S melting |
Thursday June 30
OBJECTIVES CHAPTER 11
AGENDA: CHAPTER 11 "CAUSES OF CHANGE"- THERMODYNAMICS
- Distinguish between heat and temperature
- define and calculate molar heat capacity
- cite two principals that govern most natural events
AGENDA: CHAPTER 11 "CAUSES OF CHANGE"- THERMODYNAMICS
- CLASS HAD UNPLANNED CANCELLATION. TEST TO BE EXTENDED UNTIL TUESDAY JULY 5
- When _____ is burned there is no hydrocarbons, carbon oxides are emitted. It also has 3 times more the energy content than of regular gasoline. The problem is that to be converted to a liquid to be stored results in having 3000 times less energy making the big-picture view of using hydrogen to replace petroleum-based fuels too uneconomical.
- A decrease in _____ and an increase in _____ govern chemical reactions as they do in natural events.
- Heat represents the _____ thermal energy and is measured in joules. Temperature represents the _____ of thermal energy or the average kinetic energy of matter. It does not depend upon the quantity of matter present.
- _____ is a measure of disorder in systems.
- Zero on the Kelvin scale corresponds to absolute zero or the absence of _____.
|
hydrogen
motion |
quantity
|
intensity
|
entropy
|
Wednesday June 29
AGENDA: START CHAPTER 11: THERMODYNAMICS- THE STUDY OF HEAT
- Chapter TEN, stated even in our textbook, is a chance to brush up on old skills we have learned so far. So for the first half today please work through chapter 10 and do the concept questions to make sure that you understand everything. Then turn it in for points. If you finish early you may move on to Chapter 11.
Tuesday June 28
29AGENDA:
- Obvious chemical reactions: Beautiful chemistry video (3 min 21 seconds)
- Aluminum metal in Cu(II)SO₄
- Aluminum (powder) + iodine (powder) ✑ purple vapors
- Burning magnesium ribbon
- Lead nitrate + Potassium iodide ✑ PbI₂⍗ (bright yellow ppt)
- mix baking soda and vinegar to produce CO₂ (g) HC2H3O2(aq) + NaHCO3(aq) → NaC2H3O2(aq) + H2O() + CO2(g)
- cut apple turning brown
- rusting of iron
- scrambling an egg
- milk going sour
- red & blue litmus paper in vinegar and then ammonia
- hold flame to calcium sulfate
- Some famous chemical equations: Photosynthesis, cellular respiration,
- Evidence for chemical change: heat, light, sound, gas, precipitate, color change
- Condensation, melting and crystallization are physical changes
- EXOTHERMIC (energy out) occur at room temperature OR ENDOTHERMIC (energy in) rarely occur at room temperature
- bond formation (releases energy) - g bond breaking (requires energy)
- forest fire = spontaneous exothermic reaction
- In an exothermic reacton the bonds are stronger in the product than in the reactants.
- DO ALL CONCEPT CHECKS CHAPTER NINE
- BALANCING EQUATION TIPS:
- Delay balancing the elements that occur in several reactants or products
- If the same polyatomic ions appear on both sides of the equation, treat the as single units, like monoatomic ions.
- After considering tips 1 and 2, balanions, balance the elements left to right
- Remember, balancing one element may unbalance others
- For ionic equations, be sure charges are balanced
- DIFFERENT STATES of matter: (g) for gas (s) for solid (aq) for aqueous and (l) for liquid
- COMPARE COEFFICIENTS OF BALANCED EQUATION TO GET MOLE RATIOS (mole ratios can be used as conversion factors)
- Cellular respiration: C₆H₁₂O₆(aq)+ 6O₂(g) ✑ 6CO₂(g) + 6H₂O(l) + energy............ we could use the following conversion factors:
- 1 mole glucose = 6 moles of oxygen
- 1 mole glucose = 6 moles of carbon dioxide
- 1 mole glucose = 6 moles of water
- 6 moles of oxygen = 6 moles of carbon dioxide
- 6 moles of oxygen = 6 moles of water
- 6 moles of carbon dioxide = 6 moles of water
- Energy can be written into equations as Kj (kilojoules) and can become part of a mole ratio and thus conversion unit
- Example if energy was 100 Kj value in the cellular respiration equation you could use it in a ratio as 100 Kj = 1 mole glucose = 6 moles of oxygen = 6 moles of carbon dioxide and 6 moles of water.
- Practice question: How much energy does 2 moles of glucose produce if the energy released from cellular respiration is 100 KJ?
- ANSWER: 2 moles oxygen x (100 Kj / 6 moles oxygen) = 33.3 Kj
- Example if energy was 100 Kj value in the cellular respiration equation you could use it in a ratio as 100 Kj = 1 mole glucose = 6 moles of oxygen = 6 moles of carbon dioxide and 6 moles of water.
- REACTION TYPES
- COMBUSTION: Oxygen reacts with organic compounds to produce light, heat or sound CH₄ + O₂ ✑ CO₂ + H₂O + energy
- SYNTHESIS: Complex molecules made from simpler substances: 2H₂ + O₂ ✑ 2H₂O (2 becomes 1)
- Polymerization reactions are a form of synthesis
- DECOMPOSITION: Complex molecules broken down to simpler: CH₃OH ✑ CO + 2H₂ (1 becomes 2)
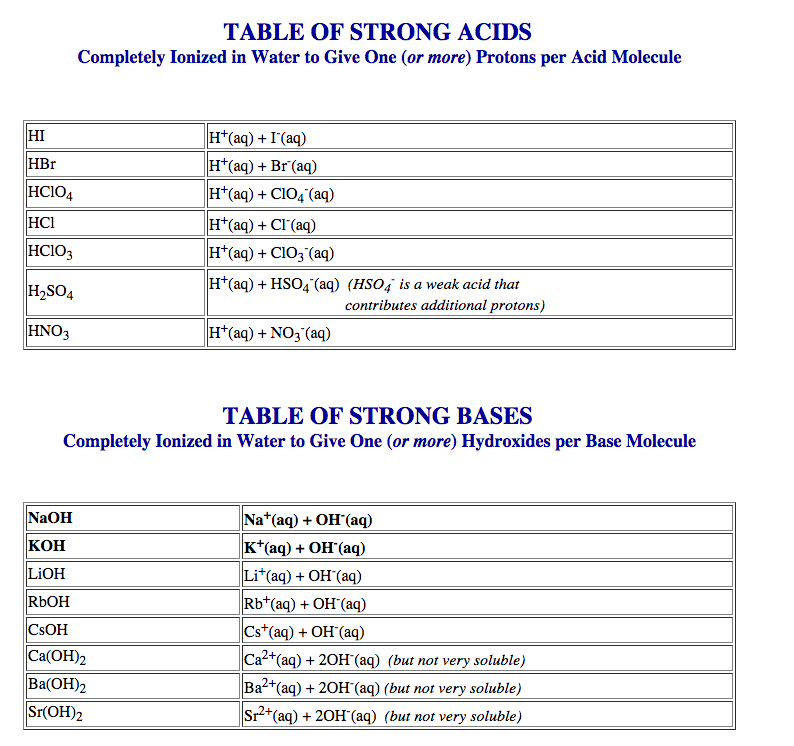
- DOUBLE DISPLACEMENT: The two cations exchange places with each other: 2KI(aq) + Pb(NO₃)₂ (aq) ✑ PbI₂(s)+ 2KNO₃(aq)
- SINGLE DISPLACEMENT: One cation displaces another: 2Al(s) + 3CuCl₂(aq) ✑ 2AlCl₃(aq) + 3Cu(s)
- Net ionic equations, spectator ions
- Double displacement reactions are more accurately written as net ionic equation since most compounds dissolve in aqueous solution so you would write the ions as such. The complete ionic equation would be: 2KI(aq) + Pb(NO₃)₂(aq) ► PbI₂(s) + 2K(NO₃)(aq) becomes... 2K+(aq) + 2I⁻(aq) + Pb²⁺(aq) + 2(NO₃)⁻(aq) ►PbI₂(s) + 2K⁺(aq) + 2(NO₃)⁻(aq)
- The spectator ions are the ones that appears on both sides of the equation but cancel each other out. If we actually remove them then we will be left with the original
- If time do test prep chapter 9 at end
- DO Concept checks for chapters 10. This chapter titles "STOICHIOMETRY" is really just making certain that students understand the following concepts: I copies this straight out the book on here
- CHAPTER 1: conversion factors
- CHAPTER 2: significant figures: density
- CHAPTER 7: nomenclature and structures of organic compounds
- CHAPTER 8: between grams and moles, molar masses, three-step method of problem solving
- CHAPTER 9: balanced chemical equations
- PAPER CHROMATOGRAPHY (To observe the separation of ink components) - 1 to each table
- Will separate the components of gas and liquid solutions by passing them through a coffee filter (porous) via capillary action by way of distilled water. The larger components will move more slowly.
- Cut filter paper into 10 X 10 cm square
- Draw a light line one cm from the edge of the paper. Starting from 1 cm from edge, make marks 2 cm apart so there will be marks at the 1, 3, 5, 7, and 9 cm points.
- On the first dot put mix of Cu/Pb(No₃)₂ solution into a well.
- Put a marker circle about 1 cm diameter on 2nd dot.
- Put food dye on third dot
- Put any other water soluble substances that you can think of for dots four and five
- Roll paper into a cylinder and fasten the edges with tape
- Put 3mm of distilled water into a 150mL beaker and put the paper into the beaker with the spots facing down
- When water is about 5mm from the top edge pull it out. This may take about 30 minutes. Lay the paper out to dry.
- Develop Cu/Pb(NO₃)₂ streak with KI cotton swab. Cu should turn brown while Pb should turn yellow and will eventually turn the copper yellow as well.
- Measure the average distance each solute (color) traveled and record this in millimeters.
- ELECTRICAL CONDUCTIVITIES: (Observe which solutions conduct electricity the best)
- Use electronic set to configure electronic conductivity tester.- 2 boxes. I have battery attached one.
- Make sugar solution (2mL table sugar) in 60mL distilled water and stir with pipet. Put in 1st well of row. (5 drops)
- Put 5 drops of HCL into 2nd well, 1st row
- Put 5 drops of NH₄OH into 3rd well, 1st row
- Put 5 drops of Copper(II) Nitrate into 4th well, 1st row
- Put 5 drops of glycerin in 5th well, 1st row
- Put 5 drops of distilled water in 6th well, 1st row.
- Bring out your conductivity apparatus
- Dip both electrodes of the tester in the first well. Was it a poor, medium, or good conductor?
- Continue to test and record the conductivity of each of the other liquids. Be sure to wash the electrodes in distilled water before each test.
- Will separate the components of gas and liquid solutions by passing them through a coffee filter (porous) via capillary action by way of distilled water. The larger components will move more slowly.
- _____ can provide the energy needed for a reaction to occur. (collisions)
- Fire extinguishers display codes indicating which types of fires they can put out. _____ is used on class A fires involving solid fuels such as wood. A class B fire, where the fuel is a liquid or gas, is best put out by _____. Class C fires involve a live wire circuit to be extinguished usually by ammonium dihydrogen phosphate or sodium hydrogen carbonate. Class D fires involve burning _____. Powders are used to cover them, like ____
- _____ rubber is very strong. Its done by joining the polymer chains together with sulfur links. It is used today to make tires, rain gear, and rubber bands.
- Polymers are also known as _____ and are made up of _____
- _____ is the most abundant natural polymer in the world.
- _____ bonds are formed between the amine ⎯N of one amino acid and the carboxylic acid group the next amino acid.
Monday June 27
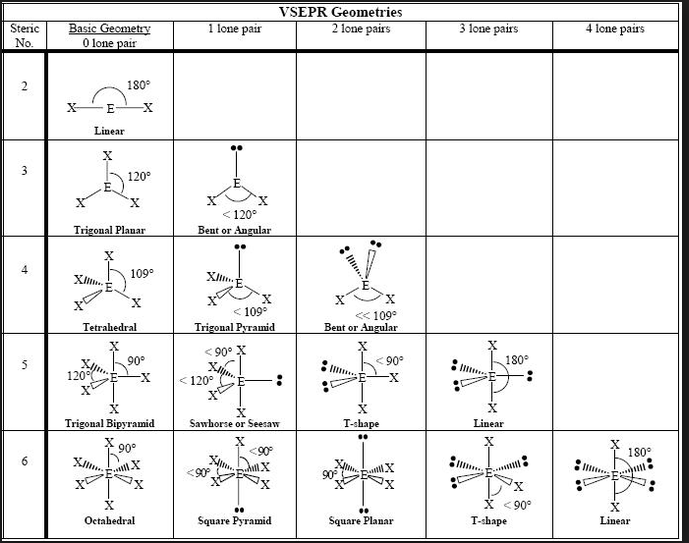
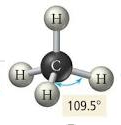
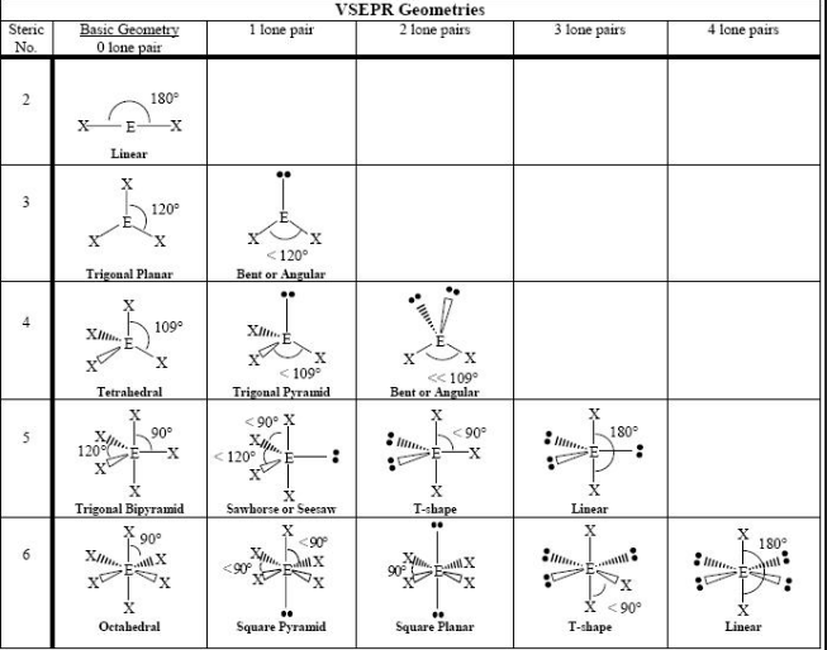
- AGENDA: Section 6-3 How are molecular shapes determined (Valence Shell Electron Pair Repulsion) theory
- After you draw your Lewis Structure then to determine molecular geometry....
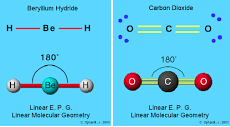
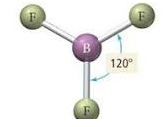
- First you obtain the number of bonds and lone electron pairs and this is your basic geometry (double & triple bonds only count as 1)
- Then you see how many lone pairs you have and look at the table from Friday (below)
- CO₂ is linear
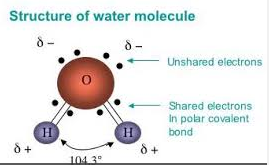
- H₂O is bent
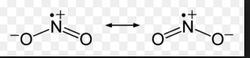
- Now practice the following: NH₂Cl & ONCl (teacher) and NO₃⁻ & NH₄⁺ (students)
- Quiz (each person will be assigned a molecule to determine the geometry of)
- In general the greater the polarity is the higher the boiling point
- Would water be polar if it had a linear shape like carbon dioxide?
- Ionic substances tend to be solids with high melting points, and molecular substances are more likely to be gases, liquids, or solids with low melting points.
- Hydrogen bonds are the strongest of the dipole-dipole and form between hydrogen and the halogens, nitrogen and oxygen.
- Lecture about 7-1 and 7-2 and leave 7-3 for students to read
- know monomers: amino acids, monosaccharides, nucleic acids, fatty acids+glycerol and what their natural polymers are called
- know alkanes, alkenes, and alkynes are single, double and triple bonds
- Students should already know 8.1
- I will do question on page 290 about empirical formulas and percentages of compounds
- Chapter 9 concept reviews
Friday June 24
AGENDA:
- Went over Lewis Structure and VSEP geometries. You don't have to memorize. I'll give you a sheet like the one below to use on tests.
- Test #3. High score was 18 so I made the 100% mark 18 and curved the class up.
Thursday June 23
AGENDA:
- Took dimensional analysis quiz again (the higher of your two scores will count)
- practiced nomenclature using the handout
- Did concept checks in chapter 6 all the way up to where VESPR began. And we stopped there. Therefore you will not be responsible for anything coming after section 6.1 and 6.2 tomorrow on the test. The test will consist of chapter 5, and 6.1 & 6.2 readings.
- Describe salt: Is it NaCl or not NaCl or is NaCl just one of many different kinds of salts?
- Know nitrate, phosphate, sulfate, and carbonate NO₃⁻ PO₄³⁻ SO₄² CO₃⁻
Wednesday June 22
AGENDA:
DEMONSTRATION (10 minutes approximately)
- PERIODIC TABLE PRACTICE
- CHALCOGENS / CHEM TEACHER ELECTRONIC AID TO GO ALONG WITH A CHEMISTRY CLASS LIKE MINE.
- Alkali metals reactions with water
- Rutherford's experiment with gold
- Brainpop: Metals, ions, chemical bonds (maybe take a quiz)
- go over nomenclature handouts and finish remainder in class
- retake dimensional analysis
- card trick (I had the 7 already folded)
DEMONSTRATION (10 minutes approximately)
- Fill a Pyrex test tube one-fourth full of table sugar (sucrose) and another test tube one-fourth ful of sodium chloride.
- Ask the students for a variety of ways to tell the two compounds apart.
- Using either a projecting microscope or several stereo or standard microscopes, have the students view the two compounds.
- Pour a little of each compound into separate 150 mL beakers that are half full of water. Check with a conductivity tester.
- Heat a nichrome wire, dip it into the sugar crystals, and place it in the flame. Reheat the wire, dip it into the salt, and place it in a flame.
- Heat both test tubes in a flame. Why did one compound melt?
- The nomenclature worksheet handout must be completed and brought to class. Be prepared for quiz.
- Chapter 6: Read it and answer all the concept checks to turn in.
- If you still are not sure about dimensional analysis you can try page 277 in text sample problems 8A and 8B are converting from moles to numbers of atoms and .... atoms of silicon to moles of silicon (examples should show you how it is worked out). As a matter of fact if you continuing reading from 278 on its its all about conversions aka "dimensional analysis" so when we get to this chapter you will find that we've covered a lot of it already. Use CHEM TEACHER WEBSITE link to help.
Tuesday June 21
AGENDA:
- Dimensional analysis followed by quiz question
- going over polyanions (-ate to -ite means having one less oxygen)
- going over binomial nomenclature- ionic compounds- metal/nonmetal with ending -ide
- The handout- bring it in to do in class with a good understanding of it having done some of it already
- Begin reading chapter six
Monday June 20
,AGENDA: TEST TODAY
- CHEMISTRY GAMES TO LEARN POLYATOMIC IONS, ACIDS, BASES, ETC...
- 4 new elements named (summer 2016 article): 113: Nihonium- Nh, 115: Moscovium- Mc, 117: Tennessine- Tn, 118: Oganesson- Og, one is a halogen and one is a noble gas.
- conversion factors (moles / molar mass) & (moles / number of atoms, molecules, ions)- QUIZ TOMORROW ON THIS
- Silver ion has charge of 1+, (in addition to GroupI)
- Cadmium, tin, platinum, zinc, nickel, cobalt, manganese, cadmium, tin, lead and lead ion has charge of 2+ (in addition to group2)
- Aluminum, gallium, ruthenium, rhodium, chromium, bismuth, indium, and thallium ions have charge of 3+
- Zirconium and hafnium ions have charge of +4
- Tantalum ion has charge of +5
- Molybdenum and Tungsten ions have charge of +6
- Nitride and phosphide ions have charge of -3
- salts are ionic compounds, and when in aqueous solution conduct electricity good. They are hard and brittle.
- Nomenclature = BINOMIAL NOMENCLATURE is the system for naming chemical substances for ionic compounds the cation (metal) is named first followed by the anion (nonmetal) which has its ending changed to -ide (e.g. NaCl = Sodium chloride)
- Common household names of compounds (Mg(OH)₂ = milk of magnesia, NaCl = table salt, NH₄OH = ammonia water, HC₂H₃O₂ = vinegar, and NaHCO₃ = baking soda)
- mention error I caught on page 165 if you tried #21 concept review. (see if anyone caught it for extra credit)
- Transition elements can have different charges and Roman numerals are used to describe them
- The term formula unit is used for ionic compounds. The term molecular formula is used for covalent compounds
- Remember the polyatomic ions written in red below (8 of them) for quiz tomorrow. But if you can memorize all from page ⁺, 171 table 5-8 that would be the best. (ions that are formed from two or more elements). If you know the -ates you can derive most of the rest.
- hypochlorite ion ClO⁻ acetate ion C₂H₃O₂⁻ carbonate ion CO₃²⁻ Phosphoric acid H₃PO₄
- chlorite ion ClO₂⁻ bromate ion BrO₃⁻ chromate ion CrO₄²⁻ ⁻ dihydrogen phosphate H₂PO₄⁻
- Chlorate ion ClO₃⁻ hydroxide ion OH⁻ dichromate ion Cr₂O₇²⁻ hydrogen phosphate HPO₄²⁻
- perchlorate ion ClO₄⁻ cyanide ion CN⁻ oxalate ion C₂O₄²⁻
- sulfate ion SO₄²⁻
- hydronium ion H₃O⁺ nitrate ion NO₃⁻
- Ammonium ion NH₄ﱣ phosphate ion PO₄³⁻
- Chlorate ion ClO₃⁻
- The only positive polyatomic ion is NH₄⁺ (ammonium ion). Another positive ion is dimercury (I) ion Hg₂²⁺. Perioxide is O₂²⁻
- There becomes one less oxygen when going from -ate to -ite (eg ClO₃⁻ to ClO₂⁻) one less than -ite is hypo- and one more than -ate is per-
- Polyatomic ions that end in -ate were derived from acids whose names ended in -ic. Thosse that end in -ite were derived from acids whose names ended in -ous. (Acids have Hd at the beginning which make up for all or part of the charge -eg- HCL
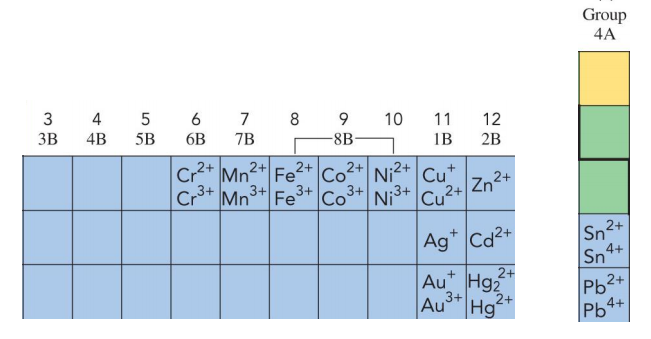
- Common Transition metal charges: Cu⁺, Cu²⁺, Cr²⁺, Cr³⁺, Cr⁴⁺, Fe²⁺, Fe³⁺, Co²⁺, Co³⁺, Ni²⁺, Ni³⁺, Sn²⁺, Sn⁴⁺, Pt²⁺, Pt⁴, Pb²⁺, Pb⁴⁺, Hg₂²⁺, Hg²⁺, Au⁺, Au³⁺
- Uses of salt: preserve food and melt ice
- ZnO is an astrigent; MgSO₄ x 12H₂O is epsom salt
- hydrated crystals are used as dessicants
| nomenclature_worksheets_covers_all.pdf | |
| File Size: | 45 kb |
| File Type: | |
Friday June 17
AGENDA: PRACTICE TEST TODAY- REAL TEST MONDAY
- Group 1 alkali metals are soft and can be cut through but very reactive (except lithium whose size pulls the electrons in)
- Group 2 alkaline earth metals are harder, denser, stronger and have higher melting points than the Group1. But even though they are reactive they are not reactive as group1s in that they must lose two rather than one electron.
- Transition metals are even harder, denser, stronger and have higher melting points than the group2 (and therefore also the group1) metals.
- The lanthanides include elements 57 to 71. All lanthanides have +3 oxidization numbers and very similar chemical propertiess
- The actinides include elements 89 to 103. All actinides have radioactive forms
- Halogens are the most reactive of the nonmetals. They combine with metals to form compounds known as salts.
- metalloids are semi-conductors but need temperature to be raised to make them conduct better while regular metals conduct electricity better as they are cooled.
Thursday June 16
AGENDA:
- Learn those elements (test yourself) game
- Phosphorous is glow-in-the-dark
- Quantum numbers in describing elements and their orbitals - 8 min (n,l,m, and s orbitals)
- Revisions to Dalton's 5 principles: In contrast to "Atoms of a given element are identical in their physical and chemical properties" we now know of isotopes whose properties, density for instance, are not identical. In contrast to "Atoms cannot be subdivided, created, or destroyed when they are combined, separated, or rearranged in chemical reactions" we have learned that atoms can be subdivided, created, and destroyed in nuclear processes.
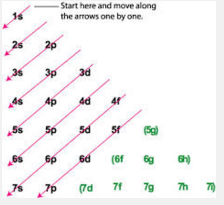
- n = principal quantum number (can be 1-7) = (energy levels)
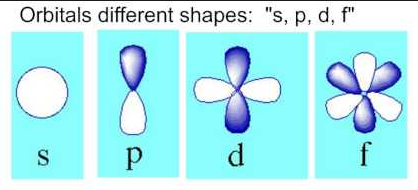
- l= the sublevel (there are the same amount of sublevels in each element as there are energy levels in its n quantum number) if l=0 then it is s orbital. If l=1 then p orbital. If l=2 then d orbital. If l=3 then f orbital.
- m = orbital. There is only 1 s orbital. There are 3 p orbitals (x,y, and z axes). There are 5 d orbitals and 7 f orbitals
- s = spin
Wednesday June 15
AGENDA:
- Test #2 this Friday will cover chapters 3 and 4
- Students completed workbook pages for chapter 4 and we started to go over the answers.
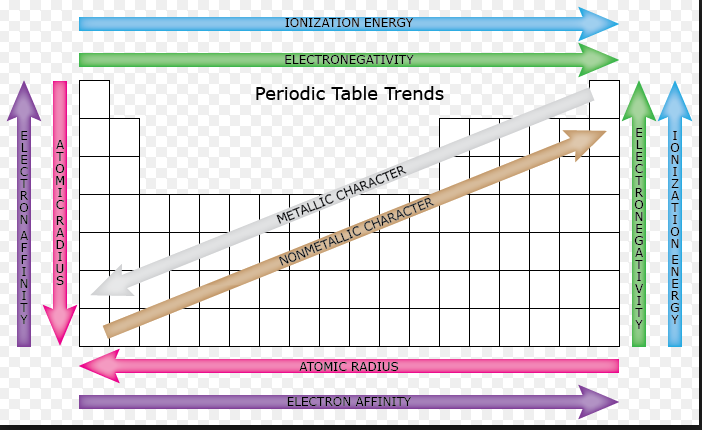
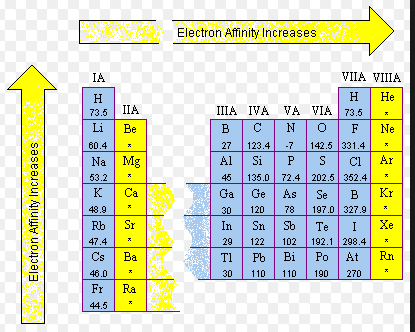
- Went over two trends: atomic radii and ionization energy which are opposites to each other:
- Atomic radii increases as you go from up to down but decreases as you move from left to right
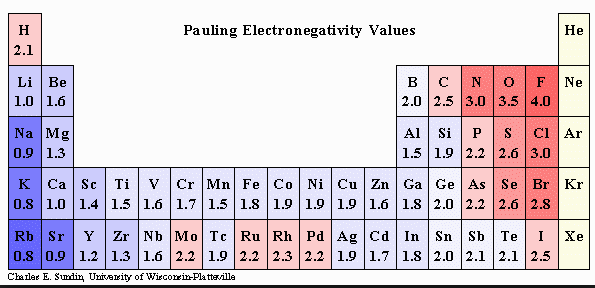
- Ionization energy has the opposite trend as atomic radii. It decreases from up to down and increases from left to right. The same trends are seen with electron affinity and electronnegativity
- orbitals explained about 10 minutes- able to picture what s and p orbitals look like a little better. It would make more sense after knowing Lewis structures which we will get to later in the course.
- n stands for the energy
- s, p, d, and f are derived from the words sharp, principal, diffuse, and fundamental. These words were first used to describe the qualities of certain spectral lines. Later it was discovered that these qualities were related to the sublevel structure of the electron arrangement, so their letters were retained.
- Chapter 3 review questions page 107 questions 19-29
- Also Chapters 2, 3, and 4 concept reviews can all be turned in by the morning of the test Friday for full credit.
Tuesday June 14
AGENDA:
- Went over all concept checks for chapter 3
- Went over most of workbook pages for chapter 3 will continue tomorrowI
- Fe is most abundant and widely used metal. Hg is only metal that is liquid at room temperature. It is used in thermometers. Titanium does not react with flesh or bones so it is used in surgical pins. Selenium increases in conductivity in response to more light shining on it. Phosphorous is used in glow-in-the-dark (phosphorescence). Carbon is the most versatile element as it joins with other atoms in many ways. Neon is a monoatomic gas that emits a brilliant red light when stimulated by electric current.
- Metals encompass the largest region of periodic table. They are lustrous, malleable and ductile and conduct electricity (Alkali are most reactive, alkaline earth, metalloids are semi-conductors, transition metals, and other metals.
- Nonmetals: do not conduct electricity nor heat well and can be ground into powder and are usually gasses at room temperature or very brittle if solid
- Start reading chapter 5 and answering concept reviews
- _____ is needed for your blood to clot when you bleed, and for your muscles to contract when you move.
- Only 100mg or less per day of _____ are required for you to stay healthy. They include, Fe, Mn, Cu, I, Zn, Co, Cr, Ni, Si, Sn, and V
- Of the more than 100 elements in the universe, 33 are essential for your health. These elements are C, H, N, and O which make up 96% of your body mass. P and S are also included if more detail is needed (CHNOPS) The P stands for _____ and the S for _____. The other major minerals, besides Ca, are K, Cl, Na, and Mg.
- Can you name the 6 metalloids? They are B, Si, Ge, As, Sb, and Te. What do they stand for? They are used to make _____ and _____
- The heaviest element in nature is _____ with atomic number of _____. Anything above this element is man-made.
- The densest element is _____ which is atomic number _____
|
uranium
Calcium 92 |
osmium
Sulfur |
76
Phosphorous semi-conductors |
trace elements
alloys |
Monday June 13
OBJECTIVES:
HOMEWORK:
- Describe organization of the periodic table; explain how the names and symbols of elements are derived; identify common metals, nonmetals, metalloids and noble gases
- Another test: take average of your two scores (19 was the high score- explain)
- The difference between acetaminophen and aspirin
- Elements also named in honor of a person, a planet, or where the element was discovered, or from older Greek and Roman words.
- Every element with a two letter symbol begins with a capital letter and is followed by a lower case letter. This way you can distinguish, for instance CO from Co.
- Elements represented by symbols containing 3 letters have not been officially named. They will be changed when the International Union of Pure and Applied Chemistry (IUPAC) gives the elements official names.
- 1 amu = 1.6605402 x 10⁻²⁴g
- PASS AROUND CHEMISTRY CARDS- EVERYBODY CHOOSE ONE AND make a flash card with the name of the element on one side and the abbreviation of the element and its region of the periodic table and some other interesting facts on the other side of the card. Then each student will show their card and first see if anyone can name it based on its symbol. Afterwards he/she will flip it over and tell the class about the element
- Example: Tungsten, which is used to make light bulb filaments, has the symbol W. It was named from the German word wolfram since it was discovered in Germany.
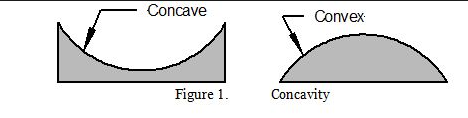
- Shape of water line in capillary tube- is called a meniscus - concave - adhesion
- The force of gravity causes raindrops to fall. They hit the ground with what kind of shape? convex - cohesion
- Students observe the glass capillary tube set. Water will be poured into the large reservoir tube and flow into the other 3 tubes.
- Students write down a prediction of the water levels in each of the 3 tubes. How is each tube different?
- I will measure water in a graduated cylinder and then will mix in food coloring. The colored water will be poured into the largest reservoir glass tube. Are the water heights the same in each tube? Students will measure and record the height of the water in each capillary tube (small, medium, and large) Is there a relationship between the height the water reaches and something else? If there seems to be then measure it and graph it. Now does the graph show a linear relationship? Is it a constant? Make a ratio between the two variables. Can you predict either the height or the other variable using a proportion?
- If you put different liquids in the capillary tubes, do they reach different heights? Why? (Rubbing alcohol , hydrogen peroxide, vinegar, etc...)
- What happens if you add a solute like sugar or salt to the water? Does the height change? Why? Explain in terms of attractive forces.
- Ready for an easy quiz using the following words? (meniscus, gravity, convex, glass, capillary action, adhesion, cohesion, hypothesis, liquid, concave)
- A downward force is _____. _____ occurs when a liquid moves upward within a glass tube. An example of a solid is _____ A water line is referred to as a _____. When a shape is lower in the middle and higher on the edges it is _____. Water is an example of a _____. _____ occurs between a solid and liquid where molecules are not like each other. When similar molecules group together, it is _____. A raindrop has a _____ shape, being higher in the middle than on the edges. A scientific prediction is a _____.
- The brilliant flashes of light produced by fireworks consist of electromagnetic radiation with _____ between 380 nm and 780 nm. They produce light through atomic and molecular _____. They also produce bright white flashed through _____. The _____ oxide particles (product) glow white hot in the intense heat generated in the reaction.
- Different elements, in elemental state or combined in compounds, produce different _____. Compounds of strontium produce _____ fireworks, aluminum compounds produce bright _____ light, barium compounds produce _____ light, and _____ atoms produce yellow light.
- How many different elements are there currently?
- The law of _____ states that a given compound contains the same elements in exactly the same proportions by mass.
- The law of _____ states that the mass of the products of a reaction equals the mass of the reactants. The famous equation states that mass and energy are equivalent
- A special mass unit is used to measure atomic mass. It is the _____ or the _____.
- A unit to serve as a bridge between the invisible world of atoms and the macroscopic world of materials and objects is the _____ which is equal to 6.0221367 x 10²³
- moles can be expressed in numbers of atoms, molecules or _____
- Elements with similar properties fall into the same group which are represented by _____ in the periodic table. All of the group I elements have low _____, low _____ points, and good electrical _____. They are ____until they react with air and are _____ enough to be cut with a knife.
- Elements in the periodic table are arranged according to increasing _____ and family trends in properties.
- The rows of the periodic table are called _____. There are _____ of them in the periodic table besides actinides and lanthanides.
- People who have a lack of _____ in their diet (for people that don't eat seafood) are much more apt to have goiters .
- Metals generally are _____ at room temperature (except _____ which is a liquid), have _____ color, a _____ surface and _____ electricity.
- _____ are used in light-sensitive devices because its conductivity increases in response to light shining on it. _____ is known for its phosphorescence, which means glowing in the dark. Argon and krypton are used in _____ lamps.
- Germanium is a semiconductor metalloid that is slightly more conductive than _____ so their circuits operate faster. Both of these elements are classified as _____
- _____ is a monoatomic gas that emits a red color.
- _____ has the lowest melting point of any element
- _____ is the best thermal conductor, is the hardest on the Mohs scale, and slows down light up to three times
- Metalloids are _____
- _____, atomic number 88, is odorless, colorless, tasteless radioactive gas emitted from underground rocks. Overexposure may cause cancer.
- _____ is the most abundant, widely used, and least expensive metal.
- _____ is the first inert gas that was found to react.
- _____ are now usually called electron beams.
HOMEWORK:
- Read chapter 3 and chapter 4 and do all the concept checks
- start learning all the rest of the elements
|
shiny
atomic number melting green incandescence silicon columns xenon |
white
metal colors conduct grayish metalloids iron conductivity |
definite proportions
radon solid periods shiny neon sodium 118 wavelengths |
semi conductors helium mercury soft density emissions red fluorescent cathode rays |
Friday June 10
TEST RESULTS:AGENDA:
- Examples of physical properties: Color, mass, texture, volume, transparency, taste, electrical conductivity, heat conductivity, melting point, boiling point, index of refraction, malleability, ductility, solubility, state of matter (solid, liquid, gas)
- Examples of chemical properties: flammability, rusting, decomposes into..., neutralizes with acid/base, milk turning sour, wood rotting, pancakes cooking, grass growing, water is absorbed by paper towel, exposure to oxygen (apple turns brown)
- kinetic energy = energy of motion; potential energy = energy due to position/location and energy in bonds
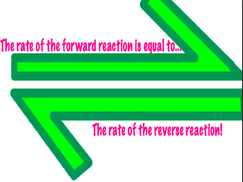
- equilibrium is when the forward rate is the same as the reverse rate. Example: dissolving
- Everything that phases down is endothermic and everything that phases up is exothermic
- Endo/Exothermic
- What are physical properties/ chemical properties
- Significant figures and scientific notation
- know all bonds and intermolecular forces
- Also it would be a good idea to have a good understanding of all the fill-in questions I have been giving on this site.
Thursday June 9
AGENDA:
HOMEWORK:
- Van der Waals forces - 4:50
- There are two kinds of Van der Waals forces: weak London Dispersion Forces and stronger dipole-dipole forces.
- Hydrogen bonding is the strongest dipole-dipole force there is.
- Intermolecular forces - 9 min (Of the intermolecular forces, all of which have less energy than covalent bonding, the hydrogen bond is the strongest. After that comes the rest of the dipole-dipole intermolecular forces (the molecules are all polar). Then there are temporary forces that occur even between non-polar molecules and these are called the London dispersion forces which are the weakest of the intermolecular forces)
- Went over carbon being 666- 6 protons, 6 neutrons, 6 electrons- and anything without any carbon in it is definitely inorganic. Proteins (made of amino acids), carbohydrates (made of monosaccharides), lipids (made of fattly acids/glycerol) and nucleic acids (made of nucleotides) are all carbon based compounds which
- Copper Copper Cube 8.9
- Brass Gold 8.0
- Steel Black Cube-Heavy 7.6
- Aluminum Silver Cube-Light 2.7
- Acrylic Clear cube 1.16 - 1.19
- Oak Thin grained cube 0.60 - 0.90
- Nylon opaque white cube 1.13
- Pine Thick Grained -Pine smell 0.35- 0.60
- Poplar Thick Grained cu 0.35 - 0.50
- PVC Grey Cube 1.39 - 1.42
HOMEWORK:
- Read Chapter Two and answer section questions
Wednesday, JUNE 8
OBJECTIVES CHAPTER ONE:
- Identify reactants and products of a chemical reaction
- Distinguish between exothermic and exothermic reactions
- Acetylsalicylic acid/aspirin; acetaminophen/Tylenol; ibuprofen/Advil (active ingredient / more commonly known names of the NSAIDs)
- While natural products are held in high esteem, there are many instances where they are too costly for widespread consumption. Example would be obtaining salicylates from willow bark. Fortunately German chemists had become experts in the development of synthetic forms of natural substances.
Tuesday JUNE 7, 2016
AGENDA:
- Hungover people everywhere, thank you Felix Hoffman - 1:10
- ionic vs covalent bonding- 1:57
- Do concept checks on page 15
- Dimensional analysis practice: "Some say that there is a hollow earth and giants that are 3 to 4 meters high reside there. Please convert 3.00 meters and 4.00 meters into normal feet and inches that we use to express someone's height." Conversion factors given: 1 foot = 12 inches; and 1 inch = 2.54 centimeters
- Start off with what is given 3.00 meters
- Now look for a possible conversion factor which has meters in it so that we cancel out the meters by putting it in the denominator. But there is no conversion factor with meters in it. However, I see one with cm which is centimeters so we can add in the following conversion factor: (100 centimeters / 1 meter)
- So now we have 3 meters x (100 centimeters / 1 meter) and we see that the meters cancel each other out. So now we look for a conversion factor with centimeters in it to cancel out the centimeters that our problem is now left with. We can use the 1 inch = 2.54 centimeters next
- So now we have 3 meters x (100 centimeters / 1 meter) x (1 inch / 2.54 centimeters) and we see that the centimeters cancel each other out and we left with the "inch" as the remaining unit.... so we use our final conversion factor (1 foot = 12 inches)
- So now we have 3 meters x (100 centimeters / 1 meter) x (1 inch / 2.54 centimeters) x (1 foot / 12 inches). And since we are left with "feet" we can end with the answer. Now use calculator and multiply all the numerator digits as well as divide by all the digits that appear in the many denominators. 3 x 100 / 2.54 / 12 = 9.84251968503937 feet
- If we were using scientific notation and significant figures we would use 3 significant figures because our given (3.00 meters) used 3 significant figures and we must use the lowest value of sig figs that we are given. So we came out with 9.84 feet and you would be correct. But lets finish this up with inches. 9.84 means over 9 but less than 10 feet. So 9 feet and how many inches?
- .84 ft x (12 inches / 1 foot) = 10.08 inches with the 2 significant figures bringing 10.08 to just 10 inches. There for something that is 3 meters in height will be 9 feet ' l0 inches tall! And that would be a giant.
- 1 cc = 1 cm³ = 1 mL =
- pH, acids & bases, sour and bitter
- Acids are the lower numbers on a pH scale where 7 is neutral. They have a high H⁺ (hyrogen ion) content and is sour. Bases are numbers higher than 7 on pH scale and the more OH⁻ (hydroxide ion) content, the more basic is the solution. Bases are bitter.
- molecular vs structural formulas- molecular formula would be H₂O for water. Its structural formula would show its bonding.j
- The only chemical elements which are stable single atom molecules at standard temperature and pressure (STP), are the noble gases. These are helium, neon, argon, krypton, xenon and radon. The heavier noble gases can form chemical compounds, but the lighter ones are non-reactive or inert. They are group 18 in your book. The halogens are group 17 in your book and consist of F, Cl, Br, I, and At. They are also non-metals and have a -1 charge. Group 16 has a -2 charge. (I am looking at the periodic table in the back of your book). Group 1 are the alkali metals and have a +1 charge. Group 2 are the alkaline earth metals and they have a +2 charge.
- Memorize the first 4 rows as well as the following specific elements: Ag (Silver), Au (gold), Sn (Tin), Cs (Cesium), Ba (Barium), Hg (Mercury), Pb (lead)
- Oxygen, phosphorus, sulfur, and carbon are allotropes (different molecular forms of an element). O₂ is diatomic oxygen gas while O₃ is ozone. They are the allotropes of oxygen just as graphite and diamond are for carbon.
- Given a specific substance and the densities of a known list of metals and metal alloys, you will need to design a procedure that will give you the data you need to determine your substance's density. With this information you should be able to tell me what your substance is made out of.
- Read chapter two and do the concept checks and section reviews for homework credit (posted @ 5:53pm)
- In the past humans relied on substances extracted from the _____ of willow trees to relieve pain and reduce fevers. Salicin was extracted from the bark and converted to a substance called _____ acid which is the active form of the pain reliever. Then sodium salicylate was made available for public use but it was irritating to the lining of the stomach. A man named Felix Hoffman was then able to synthesize another derivative which is called _____- the compound we know today as aspirin. This occurred in the late 1800s.
- Aspirin is one of the NSAIDs which stands for nonsteroidal _____ drugs.
- H₂SO₄ is the formula for _____ and is used for the production of _____, metal processing, and petroleum refining. It is the #1 produced (by weight) chemical in the United States
- _____ is one of the top five chemical producers in the United States.
- _____ is the force produced by gravity acting on mass. Scientists measure force in _____.
- When measurements are converted from one unit to another, _____ factors help to obtain the new number.
- _____ is the ratio of mass to volume.
- Liquid volume is measured with this?
- Any property of matter that can be measured without changing its chemical nature is known as a _____ property. Mass, volume, density, color, texture, melting point, and boiling point are some examples.
- Freezing and condensation are _____ physical changes. This is because heat energy is transferred from the substance to its surroundings.
- In contrast to freezing and condensation, _____ and _____ are endothermic physical changes. This is because energy in the form of heat is transferred to the substance from its surroundings.
- Whenever one or more substances are changed into new substances, a chemical change occurs. The new product will have different _____ properties which can be observed only when substances interact with one another. Evidence that a chemical change has occurred are the evolution of a _____ (you see bubbles if in liquid), the formation of a _____ and the evolution or absorption of _____ , the emission of _____, and a _____ change.
- A _____ reaction, also called a combination reaction, is one that occurs when a given compound is formed from simpler materials. Examples would be the synthesis of water from hydrogen and oxygen and the synthesis of carbon dioxide from carbon and oxygen.
- A _____ reaction (the opposite of the above) is when a compound is broken down into simpler compounds or all the way to the component elements. Examples would be the water being broken back down to hydrogen and oxygen and carbon dioxide back to carbon and oxygen.
- Terms such as light, heavy, large, and small are _____ descriptions of matter while _____ terms include numbers, which makes descriptions more exact.
- _____ is the most abundant metal on Earth and recycling it is much cheaper than producing it. It's resistance to _____ makes it suitable for many outdoor uses.
- Substances composed of bonded atoms from two or more different elements are classified as _____. There are two types of them, _____, which are stronger and ____ which are generally weaker since they have shared bonds that are not as strong. They are also classified as either _____ or _____ depending on whether or not it contains carbon.
- The smallest particle of a covalent compound that has the properties of the compound is a _____.
- An _____ is an atom or group of atoms with an electrical charge.
- _____ can be classified as homogenous or heterogenous. A _____ one contains substances that are not evenly distributed such as a salad and orange juice.
- An _____ solution is a solution in which the solvent is water.
- In water, _____ have pH values less than 7 and _____ have pH values greater than 7.
|
ion
bases ionic salicylic acid sulfuric acid density color fertilizer exothermic nitrogen gas precipitation |
qualitative
synthesis oxygen Dow Chemical graduated cylinder Monsanto corrosion molecule compounds anti-inflammatory melting |
aqueous
organic quantitative physical weight decomposition aluminum endothermic ethene Du Pont Exxon |
acids
light boiling covalent calcium oxide bark chemical conversion Hoechst Celanese heat inorganic |
SUMMER SESSION HILLSIDE H.S.&L.C.
JUNE 6, 2016 - JULY 15, 2016
FIRST DAY OF SCHOOL
AGENDA: CHAPTERS FOR THIS WEEK (1-4)
- Pre-test, syllabi, survey
- Conversion between metric units:
- Units of measure: length (meter), time (seconds), volume (liters), temperature (Kelvins or celcius), mass (gram) - mass is different than weight because weight can change depending on gravity
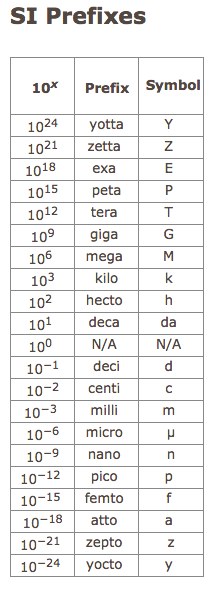
- prefixes of kilo= 1000 or 10³, milli = 1/1000 or 10⁻³ (you should have this memorized) and here are some more from 10⁻¹⁵ = femto, 10⁻¹² = pico, 10⁻⁹ = nano, 10⁻⁶ = micro, 10⁻³ = milli, 10⁻² = centi, 10³ = kilo, 10⁶ = mega, 10⁹ = giga, and 10¹² = terra
- Reactants ► Products.... Example 2H₂ + O₂ ► 2H₂O
- endothermic (needs input of energy); exothermic (gives energy usually in form heat & light)
- Acetylsalicylic acid = aspirin (for extra credit maybe- usually I will ask an extra credit question or two on a test and for the upcoming one it will probably be about aspirin since it is in your book
- Dow Chemical 2) Du Pont 3) Exxon 4) Monsanto 5) Hoechst Celanese10
- Sulfuric acid 2) nitrogen 3) oxygen 4) ethene 5) calcium oxide
CHEMISTRY RESOURCES
06/18/16 HOLT CHEMISTRY: VISUALIZING MATTER- Myers, Oldham, Tocci, Holt, Rinehart & Winston (Summer 2016 text) go.hrw.com
go.hrw.com
CHEMISTRY GAMES TO LEARN POLYATOMIC IONS, ACIDS, BASES, ETC...
CHEM TEACHER WEBSITE
all about chemistry website
General chemistry online
stoichiometry tutorials
VIRTUAL LABS
More labs
|
|
- Type in "HW0 Chapt#" with # replaced by the numbers 1 - 18 depending upon the chapter you are on. For the www.scilinks.org website you can visit the following accompanying topic pages by typing in the following keywords:
|
Chemical energy: HW021
Physical/Chemical Change: HW022 Water properties Helium: HW023 Chance Discoveries: HWl024 |
06/01/16 Holt, Rinehart & Winston Modern Chemistry Resources (just another text reference)
06/01/16 Holt Chemistry (just another text reference)
08/12/15 Online companion to Chemistry book (any time that you see this gold/orange color in this website that means it is a link that can be "clicked" on). This goes with the text "WORLD OF CHEMISTRY MCDOUGAL LITTELL- By: Zumdahl, Zumdahl & DeCoste) (Used for 2015-2016 school year)
03/02 Chemical demonstrations from MIT videos
03/03 High school chemical demonstrations from about.com videos
03/04 Motivate students with chemistry demos- Carolina reading
11/20 Chemistry games: elements, polyanions, ionic vs molecular bonds, weak/strong/nonacids vs weak/strong/nonbases, etc...
11/20 PhET Simulations General Chemistry
09/27 The Periodic Table of Elements
10/21 High School Chemistry
09/27 Widipedia Chemistry Portal
09/27 General Chemistry Online
08/12 Browse chemistry subjects ck-12
03/02 ck-12 chemistry practice09/27 Soichiometry free course
09/27 Chemistry Free Text Openstax
08/12 Here is a whole collection of chemistry videos for each chapter in the book
08/14 All the elements in cartoon character design
08/17 chemical elements https://en.wikipedia.org/wiki/Chemical_element
09/30 FunScienceDemos (good youtube demo videos)
02/01 Crash course in chemistry (46 videos)
Chemcollective virtual lab
Everything about education (everything you need about all the subjects)
Element Balancing Game
Element Concentration
chemistry4kids
Element Crossword Puzzles
Element Flash Cards
Element Hangman
Element Matching Game
Element Math Game
Element Word Scramble
08/26 Virtual Chemistry Simulations
Element BINGO
08/28 Statue of liberty original color was not green (property of copper)
08/28 about alloys
09/02 The particle Adventure (quarks and leptons, etc... )
09/02 CK-12 Plix Chemistry
03/02 ck-12 Plix chemistry
03/02 ck-12 chemistry videos
10/09 Chemistry Categories
10/09 Chemistry science demonstrations, experiments and projects
10/09 Chemistry Basics
ASTRONOMY WEB LINK from Griffith Observatory
Chemical Elements from Griffith Observatory
PRE VISIT ACTIVITIES - Griffith Observatory
http://www.NeoK12.com and username is tsanelahachi and password is 0808jtr
Pearson Chemistry Homepage for text
Chemistry animations from Learners TV
Chemistry links given to me by Daniel
http://intro.chem.okstate.edu/1314F00/Lecture/Chapter10/VSEPR.html - Molecular Geometry (for organic chemistry) VSEPR
http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch8/table.html - more VSEPR for shape depending on valence electron numbers
http://www.chemteam.info/Mole/Avogadro-Number-CalcsII.html - moles and Avagadro's number
http://www.chemteam.info/Mole/Mole.html - The Mole
http://chemistry.about.com/cs/chemistry101/a/aa071503a.htm - Reactions in water or aqueous solutions
http://chemistry.bd.psu.edu/jircitano/aqueous.html - problems with Precipitation, Acid-Base and RedOx reactions
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Reactions_in_Aqueous_Solutions/Precipitation_Reactions - solubility rules for ppt reactions
http://www.chem.sc.edu/faculty/morgan/resources/solubility/ - more solubility rules
http://www.ausetute.com.au/solrules.html - solubility table
http://www2.ucdsb.on.ca/tiss/stretton/CHEM1/stoich2.html
http://chemistry.about.com/od/chemicalreactions/a/oxidation-reduction-reactions.htm
http://www.iun.edu/~cpanhd/C101webnotes/chemical%20reactions/combustion.html
http://www.chemteam.info/Equations/Synthesis.html
https://chemfiesta.wordpress.com
06/01/16 Holt Chemistry (just another text reference)
08/12/15 Online companion to Chemistry book (any time that you see this gold/orange color in this website that means it is a link that can be "clicked" on). This goes with the text "WORLD OF CHEMISTRY MCDOUGAL LITTELL- By: Zumdahl, Zumdahl & DeCoste) (Used for 2015-2016 school year)
- Chemistry: Matter and Change © 2008
- Chemistry: Matter and Change © 2005
- Chemistry: Matter and Change © 2002
- Merrill Chemistry
- Chemistry: Concepts and Applications © 2009
- Chemistry: Concepts and Applications © 2005
- Chemistry: Concepts and Applications © 2002
03/02 Chemical demonstrations from MIT videos
03/03 High school chemical demonstrations from about.com videos
03/04 Motivate students with chemistry demos- Carolina reading
11/20 Chemistry games: elements, polyanions, ionic vs molecular bonds, weak/strong/nonacids vs weak/strong/nonbases, etc...
11/20 PhET Simulations General Chemistry
09/27 The Periodic Table of Elements
10/21 High School Chemistry
09/27 Widipedia Chemistry Portal
09/27 General Chemistry Online
08/12 Browse chemistry subjects ck-12
03/02 ck-12 chemistry practice09/27 Soichiometry free course
09/27 Chemistry Free Text Openstax
08/12 Here is a whole collection of chemistry videos for each chapter in the book
08/14 All the elements in cartoon character design
08/17 chemical elements https://en.wikipedia.org/wiki/Chemical_element
09/30 FunScienceDemos (good youtube demo videos)
02/01 Crash course in chemistry (46 videos)
Chemcollective virtual lab
Everything about education (everything you need about all the subjects)
Element Balancing Game
Element Concentration
chemistry4kids
Element Crossword Puzzles
Element Flash Cards
Element Hangman
Element Matching Game
Element Math Game
Element Word Scramble
08/26 Virtual Chemistry Simulations
Element BINGO
08/28 Statue of liberty original color was not green (property of copper)
08/28 about alloys
09/02 The particle Adventure (quarks and leptons, etc... )
09/02 CK-12 Plix Chemistry
03/02 ck-12 Plix chemistry
03/02 ck-12 chemistry videos
10/09 Chemistry Categories
10/09 Chemistry science demonstrations, experiments and projects
10/09 Chemistry Basics
- Chemistry: Matter and Change © 2008
- Chemistry: Matter and Change © 2005
- Chemistry: Matter and Change © 2002
- Merrill Chemistry
- Chemistry: Concepts and Applications © 2009
- Chemistry: Concepts and Applications © 2005
- Chemistry: Concepts and Applications © 2002
ASTRONOMY WEB LINK from Griffith Observatory
Chemical Elements from Griffith Observatory
PRE VISIT ACTIVITIES - Griffith Observatory
http://www.NeoK12.com and username is tsanelahachi and password is 0808jtr
Pearson Chemistry Homepage for text
Chemistry animations from Learners TV
Chemistry links given to me by Daniel
http://intro.chem.okstate.edu/1314F00/Lecture/Chapter10/VSEPR.html - Molecular Geometry (for organic chemistry) VSEPR
http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch8/table.html - more VSEPR for shape depending on valence electron numbers
http://www.chemteam.info/Mole/Avogadro-Number-CalcsII.html - moles and Avagadro's number
http://www.chemteam.info/Mole/Mole.html - The Mole
http://chemistry.about.com/cs/chemistry101/a/aa071503a.htm - Reactions in water or aqueous solutions
http://chemistry.bd.psu.edu/jircitano/aqueous.html - problems with Precipitation, Acid-Base and RedOx reactions
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Reactions_in_Aqueous_Solutions/Precipitation_Reactions - solubility rules for ppt reactions
http://www.chem.sc.edu/faculty/morgan/resources/solubility/ - more solubility rules
http://www.ausetute.com.au/solrules.html - solubility table
http://www2.ucdsb.on.ca/tiss/stretton/CHEM1/stoich2.html
http://chemistry.about.com/od/chemicalreactions/a/oxidation-reduction-reactions.htm
http://www.iun.edu/~cpanhd/C101webnotes/chemical%20reactions/combustion.html
http://www.chemteam.info/Equations/Synthesis.html
https://chemfiesta.wordpress.com
|
**CHEMISTRY EXPERIMENTS **
Alkali metals react explosively w/water - 3:10 Brainpop: Chemlab: Ph &* meter calibration |
SECTION QUIZZES
INTERESTING CHEMISTRY TOPICS
|
- VISIONLEARNING CHEMISTRY: INTERACTIVE ANIMATION ⎆
- CHEMISTRY BRAINPOPS: Acids and Bases, agricultural revolution, air pollution, algae, allergies, alzheimer's disease, anthrax, antibiotic resistance, atomic model, atoms, bacteria, balance, batteries, benjamin franklin, biofuels, blood, body chemistry, carbohydrates, carbon cycle, carbon dating, cellular respiration, chemical bonds, chemical equations, compounds and mixtures, conservation of mass, conserving energy, converting fractions to decimals, crystals, current electricity, customary units, decimals, diffusion, electricity, electromagnetic spectrum, electromagnets, energy sources, estimating, exponents, fats, fire, fireworks, forms of energy, fossil fuels, fractions, fuel cells, gas & oil, headaches, independent and dependent events, industrial revolution, inequalities, ions, isaac newton, isotopes, kinetic energy kurt vonnegut, marie curie, matter changing states, measuring matter, metabolism, metals, metric units, metric vs. customary, microscopes, mineral identification, moles, multiplying decimals, multiplying and dividing exponents, multiplying and dividing fractions, nanotechnology, natural resources, neurons, nikola tesla, nitrogen cycle, nobel prize, nuclear energy, nutrition, obesity, organic food, ozone layer, pasteurization, peak flow meter, percents, period, periodic table of elements, photosynthesis, plastic, potential energy, precision & accuracy, property changes, proportions, radar, radioactivity, rainbows, ratios, rounding, salt, science projects, scientific method, solar energy, standard & scientific notation, states of matter, static electricity, sun, temperature, thomas edison, thunderstorms, using a calculator, water, pH scale
August 12
First day of school
2015-2016
Agenda
Introduction to course (go over syllabus)
Personal Survey (dropbox not working this morning)
Homework: Have parents go to this website http:www.jeremyrosen.weebly.com, follow the links to get to the syllabus for the course that you are enrolled in. Then have them find my email which is listed on the cover page (so that they will have it and be able to contact me in the future- [email protected]) and have them email me a confirmation that they have seen the syllabus of the course (and have also just helped you get credit for your first homework assignment). If they have any suggestions or concerns they may address their issues during this initial email.
Introduction to course (go over syllabus)
Personal Survey (dropbox not working this morning)
Homework: Have parents go to this website http:www.jeremyrosen.weebly.com, follow the links to get to the syllabus for the course that you are enrolled in. Then have them find my email which is listed on the cover page (so that they will have it and be able to contact me in the future- [email protected]) and have them email me a confirmation that they have seen the syllabus of the course (and have also just helped you get credit for your first homework assignment). If they have any suggestions or concerns they may address their issues during this initial email.
| chemistry_syllabus.pdf | |
| File Size: | 95 kb |
| File Type: | |
| mr_rosen_survey_questions.pdf | |
| File Size: | 48 kb |
| File Type: | |
Thursday August 13
Objective(s):
Agenda
Liar game - everyone goes to the board at least once
mention the surveys are posted now
Go through beginning of book to see what people know and explain that everything they are expected to know will be posted
Mention extra credit for anyone who brings in any good chemistry videos, news, articles, websites, etc
Show links at top
Discuss
independent and dependent variables
Metric system: length is meter, mass kilogram, volume is liter , time is seconds, temperature is Celcius,
Scientific Method: Observation, hypothesis, experiment, conclusion
mass always stays the same whereas the weight of an object changes
- To understand the importance of learning chemistry
- To define chemistry
Agenda
Liar game - everyone goes to the board at least once
mention the surveys are posted now
Go through beginning of book to see what people know and explain that everything they are expected to know will be posted
Mention extra credit for anyone who brings in any good chemistry videos, news, articles, websites, etc
Show links at top
Discuss
independent and dependent variables
Metric system: length is meter, mass kilogram, volume is liter , time is seconds, temperature is Celcius,
Scientific Method: Observation, hypothesis, experiment, conclusion
mass always stays the same whereas the weight of an object changes
Friday August 14
Question
Agenda
- Make an observation (1st step in scientific process) about anything in this classroom and then do the create a question out of it. Then try to come up with some kind of question that can be answered. Explain to me what steps 2, 3, and 4 (so you are giving me an example of the scientific method carried out)
- How many chemical elements can you name?
Agenda
- Your going to pick an elements and tell the class about it. You can use your computers to find out information. No two people doing the same element. If you click here and then click on the element you will find more than enough information about it as far as physical and chemical properties. But I also want to see fun facts, the history of the element, how was it found, etc...
Monday August 17
Objective(s):
Questions
Agenda: CHAPTER TWO: MATTER
Discussion
Homework
- To learn about the composition of matter
- To learn the difference between elements and compounds
- To define the three states of matter
Questions
- In your own words what is an isotope?
- What can you say for sure about element #5?
- Which takes up more mass, 1 kg of feathers or 1 kg of steel?
Agenda: CHAPTER TWO: MATTER
- Finish element activity
- know difference mixtures and solutions
- know difference between physical and chemical properties
Discussion
- A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules arecompounds.
- A law tells us what happens; a theory (model) is our attempt to explain why it happens
- chem movies: love potion #9, project, white phosphorous, o
Homework
- Start to read chapter two
Tuesday August 18
Questions:
Agenda:
Discuss:
- What element does the symbol Pb stand for?
- What element is pencil lead made out of?
- What did lead used to be in but was taken out of and thats why its name still has the word "lead" in it?
- How come some of the symbols for the elements have letters that do not even appear in the element name like #1 above?
- Name a couple of things that you learned about specific elements yesterday (from yours and the elements that others did)
Agenda:
- PRE-TEST (30 QUESTIONS MULTIPLE CHOICE) click on LINK
- To learn the names of some of the elements
Discuss:
- An inverse relationship between the level of cobalt in a prisoner's body and the degree of violence in his behavior was found
- Alumininum exposure in water purification, cookware and utensils have indications that we should limit the intake of them
- fluoride in water supplies and toothpaste are thought to be harmful. Fluoride damages the pineal gland.
- Lithium is considered to be a miracle drug for manid-depressive aka "bi-polar" patients
Wednesday August 19
Questions:
Agenda:
- Are all mixtures solutions or are all solutions mixtures?
- Are all molecules compounds or are all compouds molecules?
- Is a solution a homogeneous or heterogenous mixture?
- Name a homogeneous mixture. Name a heterogenous mixture
Agenda:
- Play Chemistry 1 Video. (1 hour 18 min)
- PROBING MATTER (QUESTIONS): Can make game out of it - baseball - Four categories: 1) Mixtures & compounds 2) Physical & Chemical changes 3) Properties of Matter and 4) The Periodic Table
- A physical change involves a change in one or more physical properties, but no change in the fundamental components that make up the substance. The most common physcial changes are changes of state: solid - liquid - gas.
- A chemical change involves a change in the fundamental components of the substance; a given substance changes into different substance or substances. Chemical changes are called reactions: silver tarnishes by reacting with substances in the air; a plant forms leaf by combining various substances from the air and soi; and so on.
Thursday August 20
Questions
Agenda
Discussion
- Explain the difference between atoms, compounds and elements
- What are some chemical properties of a substance?
- What are some physical properties of a substance?
- What is the difference between molecules and compounds?
- What is the difference between mixtures and solutions?
- Is the air around us a solution?
- What is another name for a solution? (hint: what kind of mixture is it?)
- 24 karat gold is an element, true or flase?
- 18 karat and 14 karat gold are not elements, they are called what?
Agenda
- Chemistry 1 video VHS
- Review (Test planned to be on Monday)
Discussion
- If the atom were expanded to the size of a huge stadium like the Astrodome, the nucleus would be only about as big as a fly at the center
- if the nucleus were the size of a grape, the radius of the atom would be about one mile
- the chemistry of an atom arises from its electrons
- Atomic number: the number of protons
- Atomic mass: the sum of the numbers of protons and neutrons
- Isotopes are "top heavy": that is, the mass number is on the top top and the atomic number s on the bottom
Friday August 21
Question(s)
Discussion
- Describe how you would find the density of an object using all the tools that we have in the classroom
- What happens during a distillation?
- What happens during a filtration? (Note to self: film)
- Chapter 1 vocab (flip cards)
- Chapter 2 vocab (flip cards)
- Electrolysis of water
- Chemistry Chapters 1 & 2 audio review
- You should know the 2 different ways to separate mixtures - distillation and filtration
- Try to do the end of chapter review questions with the remainder of the time and I will assist you with the ones you get stuck on.
- Best way to review is to make sure you know all the questions up until this date in my website. Then make sure you've read what I covered under discussions and the agenda. Then I would go over all the chapter section reviews in the book and see if you can answer them as well as the end of chapter assessment. After all that you can go to the Online companion to Chemistry book and click on "assessments" and take all of them under chapters 1 & 2 and most of the questions I put on my tests now and in the future will mostly taken rom all these places just described. And it is better to start doing these chapter section assessments earlier on throughout the course so that if you are stuck with one you can ask me about it the next day. When its the last day its too late already.
Discussion
Vocabulary chapters 1 & 2
|
VOCABULARY
Chemistry theory scientific method measurement solution heterogeneous mixture distillation filtration |
matter
atom compound molecule element graphite diamond buckminsterfullerene solid |
liquid
gas physical properties chemical properties reaction mixture alloy pure substance homogeneous mixture |
THE VOCABULARY AS WELL AS THE SUMMARY FOR BOTH CHAPTERS ONE AND TWO CAN BE FOUND AT THE END OF EACH CHAPTER
Monday August 24
Objective(s):
Agenda: CHAPTER THREE: CHEMICAL FOUNDATIONS: ELEMENTS, ATOMS AND IONS
Advice: One of the major adjustments that most students have to make in going from high school to college is the need to be much more independent learners, especially by using textbooks. Students who never learned to use textbooks effectively in high school have a much more difficult time adjusting to college. Students who are not college bound also need to know how to learn from books since changing job markets necessitate retraining.
- To continue to learn facts about some elements and also learn the symbols of the elements
- Understand the law of constant composition
- Learn how a formula describes a compound's composition
- Learn the properties of metals, nonmetals, and metalloids
- Describe the formation of ions from their parent atoms
- Describe how ions combine to form neutral compounds
- To learn about the terms isotope, atomic number and mass number and to understand the A/Z(X) symbol to describe a given atom.
Agenda: CHAPTER THREE: CHEMICAL FOUNDATIONS: ELEMENTS, ATOMS AND IONS
- Test on chapters 1 and 2
- Start reading chapter 3 (read up to section 3.3 and be ready to answer the focus section questions tomorrow)
- Remember if you need extra help you can go to https://www.khanacademy.org for extra help
Advice: One of the major adjustments that most students have to make in going from high school to college is the need to be much more independent learners, especially by using textbooks. Students who never learned to use textbooks effectively in high school have a much more difficult time adjusting to college. Students who are not college bound also need to know how to learn from books since changing job markets necessitate retraining.
|
VOCABULARY CHAPTER THREE
element symbol law of constant composition atom compound chemical formula electron nucleus ion cation |
proton
neutron isotopes atomic number mass number periodic table groups alkali metals anion |
alkaline earth metals
halogens noble gases transition metals metals nonmetals metalloids (semimetals) diatomic molecule ionic compound |
CHEMISTRY TEST #1
1) A quantitative observation is known as a(n)
(A) theory
(B) measurement
(C) hypothesis
(D) experiment
2) Observations may be _____. (Choose the best answer.)
(A) quantitative
(B) neither quantitative nor qualitative
(C) qualitative
(D) both quantitative and qualitative
3) Diamond is a(n) _____.
(A) element
(B) compound
(C) homogeneous mixture (solution)
(D) heterogeneous mixture
(E) energy
4) Heat is a type of _____.
(A) element
(B) compound
(C) homogeneous mixture (solution)
(D) heterogeneous mixture
(E) energy
5) H20 aka water is which of the following
(A) element
(B) compound
(C) molecule
(D) b and c
6) Freezing point is a physical or chemical property?
7) Tooth decay physical or chemical property?
8) Flammability is a physical or chemical property?
9) Bowling balls are shiny. This is qualitative or quantitative data?
10) list the scientific method in order (4 steps)
11) Is a molecule a specific type of compound or vice versa?
12) Groups are the names of the elements that go from up to down on the periodic table. What are the names of the elements that go from right to left?
13) Another name for a homogenous mixture is what?
14) Which one is true?
a) 24 karat gold is top notch jewelry
b) 24 karat gold is an alloy
c) 14 and 18 karat gold are alloys
d) 14, 18, and 24 karat gold are all alloys
15) What is the atomic mass?
a) the number of protons in an element only
b) the number of protons, neutrons and electrons in an element
c) the number of protons and neutrons in an element
d) the mass (in grams) of all the protons and neutrons in an element.
(A) theory
(B) measurement
(C) hypothesis
(D) experiment
2) Observations may be _____. (Choose the best answer.)
(A) quantitative
(B) neither quantitative nor qualitative
(C) qualitative
(D) both quantitative and qualitative
3) Diamond is a(n) _____.
(A) element
(B) compound
(C) homogeneous mixture (solution)
(D) heterogeneous mixture
(E) energy
4) Heat is a type of _____.
(A) element
(B) compound
(C) homogeneous mixture (solution)
(D) heterogeneous mixture
(E) energy
5) H20 aka water is which of the following
(A) element
(B) compound
(C) molecule
(D) b and c
6) Freezing point is a physical or chemical property?
7) Tooth decay physical or chemical property?
8) Flammability is a physical or chemical property?
9) Bowling balls are shiny. This is qualitative or quantitative data?
10) list the scientific method in order (4 steps)
11) Is a molecule a specific type of compound or vice versa?
12) Groups are the names of the elements that go from up to down on the periodic table. What are the names of the elements that go from right to left?
13) Another name for a homogenous mixture is what?
14) Which one is true?
a) 24 karat gold is top notch jewelry
b) 24 karat gold is an alloy
c) 14 and 18 karat gold are alloys
d) 14, 18, and 24 karat gold are all alloys
15) What is the atomic mass?
a) the number of protons in an element only
b) the number of protons, neutrons and electrons in an element
c) the number of protons and neutrons in an element
d) the mass (in grams) of all the protons and neutrons in an element.
Wednesday August 26
AGENDA:
- First time with block schedule
- 1st laboratory - EXPLORING THE LABORATRY, signing safety sheet, go over names of general lab equipment and safety devices; e.g. . . which one is the Erlenmeyer flask? the graduated cylinder? lab books explained (also see syllabus)
- PROCESS OF DIFFUSION USING PLAIN TAP WATER AND FOOD DYE (1ST trial = regular tap water, 2nd trial performed mixing, 3rd trial - suppose there was heated water (could not find bunsen burners but will be available for next lab) what would the observation look like then. Students to write down exactly the observations and differences between the 3 and then make a conclusion afterwards as to why the difference.
- ELECTROLYSIS OF WATER- extra credit- set up the experiment and prepare the salt solution as described on the instructions that I will have available. Complete a circuit using the following: the salt, balance, test tubes, electrodes, and 9-volt batteries. The experiment was a success and one could see the two different gasses (H2) and (O2) being bubbled up from the water into their respective holders (tubes placed upside down in the aqueous solution) - CONGRATULATIONS ANDREW NICHOLS FOR COMPLETING THIS EXPERIMENT IN ADDITION TO FOLLOWING ALONG WITH THE REST OF THE DEMONSTRATIONS.
Friday August 28
AGENDA:
DISCUSSION:
HOMEWORK:
- chapter three
DISCUSSION:
- An independent variable is the factor that affects the outcome of the experiment. It is not affected by the experiment. Time is ofte an independent variable The dependent variable results from changes to the independent variable in the experiment.
HOMEWORK:
- Write a summary about the documentary that you saw and what it was about. What does Alternative Three mean? (This should be one-half page). Then fill in at least another quarter page with your opinion of what we watched.
Monday August 31
QUESTIONS:
AGENDA (always includes questions although does not list it. The agenda starts after going over the questions above):
DISCUSSION:
- Name the top 6 elements that are most abundant in the human body.
- How many elements are known today and how many of those occur naturally?
- Trace elements = small but crucial" elements involved with behavior and body functioning. Here are some Trace Elements in the Human Body: arsenic, chromium, cobalt, copper, fluorine, iodine, manganese, molybdenum, nickel, selenium, silicon, lithium, and vanadium Pick 2 and tell me their importance.
- Exposure to certain elements may be harmful. Describe an example.
- Not supposed to know yet: Rutherford directed alpha particles (helium nucleus) toward a thin metal foil. Most passed straight through the foil, as expected But some of the particles were deflected at large angles. What did this imply?
AGENDA (always includes questions although does not list it. The agenda starts after going over the questions above):
- Go over Focus questions 3.1 - 3.3
- Homework due: Documentary summary (1/2 page) and opinion (1/4 page)
- Go over retesting criteria (this time only I will take the better of the two test scores. But from now on I will take the "average" of the two test scores so that gives you more assentive to do good the first time.
- You are going to be responsible to learn the symbols for the elements. The only way to learn that I know of, unfortunately, is to memorize them. The following are easily confused: Au/Ag, Na/S, K/P, and Mg/Mn. What are some elements named after a place? What are some elements named after a planet? What are some elements named after a person? What are some elements with single-letter symbols?
- Have students go up to the board and draw a representation of a formula that I write up. Don't worry about its shape. Focus on the number and kind of atoms that are in the molecules. (N2O, NH3, CO2, and CH4)
- Have student go and write on board the formula for each of the following compounds: 1) A molecule contains 4 phosphorus atoms and ten oxygen atoms. 2) A molecule contains one uranium atom and six fluorine atoms.
DISCUSSION:
- Certain molecules, such as CO2, CH4, and N2O, strongly absorb infrared light. This causes the earth's atmosphere to retain more of its energy.
- Focus questions sections 3.4 - 3.7
Tuesday September 1
QUESTIONS:
- Was it JJ or Lord Kelvin who had the last name of Thomson and which one of them discovered the electron? How?
- What did the other (from 1 above) contribute to science?
- Which does actually have the larger mass if you really want to get detailed about it, the proton or neutron?
- What do you think the neutron does or is it just a thing that has mass that doesn't do anything?
- Using Z,A and X draw the symbol for an atom.
- Give the number of protons, neutrons, and electrons in the atom symbolized by 90/38SR. And 201/80Hg.
- Write the symbol for the silver atom (Z=47) that has 61 neutrons
- Answer questions and then rest of the time will be doing the retake for test #1
- about alloys
- Read sections 3.8 and 3.9. Try all practice and examole problems
- Focus question 3.8 - 3.11 due Thursday
Wednesday September 2
AGENDA:
STUDY AIDS:
TEST #1 RETAKE
TEST #1 RETAKE
1) Give me an example of a qualitative observation.
2) Give me an example of a quantiative observation.
3) Diamond (containing atoms of Carbon only) is a (n) _____.
(A) element
(B) compound
(C) neither a and b
(D) both a and b
4) Heat is a type of _____.
(A) element
(B) compound
(C) homogeneous mixture (solution)
(D) heterogeneous mixture
(E) energy
5) H20 aka water is which of the following
(A) element
(B) compound
(C) molecule
(D) b and c
6) Melting point is a physical or chemical property or both?
7) Tooth decay physical or chemical property?
8) The process of cooking foods involves physical or chemical properties?
9) Bowling balls are shiny. This is qualitative or quantitative data?
10) What part of the scientific method leads to questioning?
11) What is the name of a specific type of compound that has covalent bonds?
12) Groups are the names of the elements that go from up to down on the periodic table. What are the names of the elements that go from right to left?
13) Another name for a homogenous mixture is what?
14) Which one is true?
a) 24 karat gold is top notch jewelry
b) 24 karat gold is an alloy
c) 14 and 18 karat gold are alloys
d) 14, 18, and 24 karat gold are all alloys
15) What is the atomic mass?
a) the number of protons in an element only
b) the number of protons, neutrons and electrons in an element
c) the number of protons and neutrons in an element
d) the mass (in grams) of all the protons and neutrons in an element.
- Reminders for what is due: syllabus, survey, alternative summary, keeping lab book & questions
- RETAKE TEST
- kahoot.it
- Formation of ionic compound animation
- Comparison of molecular and ionic compound animation
- Gold foil experiment animation
- Cathode ray tube video
- Covalent vs Ionic bonding - simulation
- Solids, liquids and gas concepts
- Types of bonds problem sheet
STUDY AIDS:
- Elements and atoms video - 13:09
- Chemical symbols of elements video - 1:23
- Compangn types of bonds (overview) - 1:34
- Comparing types of bonds (example 1) - 2:20
- 3 different videos about solids, liquids and gases
- Focus question 3.8 - 3.11 due Thursday
TEST #1 RETAKE
TEST #1 RETAKE
1) Give me an example of a qualitative observation.
2) Give me an example of a quantiative observation.
3) Diamond (containing atoms of Carbon only) is a (n) _____.
(A) element
(B) compound
(C) neither a and b
(D) both a and b
4) Heat is a type of _____.
(A) element
(B) compound
(C) homogeneous mixture (solution)
(D) heterogeneous mixture
(E) energy
5) H20 aka water is which of the following
(A) element
(B) compound
(C) molecule
(D) b and c
6) Melting point is a physical or chemical property or both?
7) Tooth decay physical or chemical property?
8) The process of cooking foods involves physical or chemical properties?
9) Bowling balls are shiny. This is qualitative or quantitative data?
10) What part of the scientific method leads to questioning?
11) What is the name of a specific type of compound that has covalent bonds?
12) Groups are the names of the elements that go from up to down on the periodic table. What are the names of the elements that go from right to left?
13) Another name for a homogenous mixture is what?
14) Which one is true?
a) 24 karat gold is top notch jewelry
b) 24 karat gold is an alloy
c) 14 and 18 karat gold are alloys
d) 14, 18, and 24 karat gold are all alloys
15) What is the atomic mass?
a) the number of protons in an element only
b) the number of protons, neutrons and electrons in an element
c) the number of protons and neutrons in an element
d) the mass (in grams) of all the protons and neutrons in an element.
Friday September 4
AGENDA:
- Go over the retake test
- Go over everything that we have done in this chapter and review
Tuesday September 8
QUESTIONS:
- Write the following compounds: A molecule containing six carbon atoms and six hydrogen atoms; A compound containing equal numbers of sodium and nitrogen atoms, but three times as many oxygen atoms as there are sodium atoms
- Determine the number of each of the three types of subatomic particles for each of the following: 60/27Co; 238/92U
- Give the symbol and atomic number and specify whether the element is a metal or nonmetal and the name of the family to which it belongs: Strontium, silicon, xenon
- Use the pairs of ions below to give the formula for the compound containing these ions: Ba2+ and O2-; Al3+ and S2+; K+ and P3-
- Practice questions on the board, take turns with students
- know the symbols for elements 1 - 20, all elements in groups 1,2, 6,7 and 8, and all the ones I named in class such as Co, Cu, Zn, As, Ag, Au, Sn, Pb, Fe, Hg (cobalt, copper, zinc, arsenic, silver, gold, tin, lead, mercury)
- Focus Questions 3.8 to 3.11 and start studying the queations at the end of the chapter
Wednesday September 9
QUESTIONS:
- Which of the following are examples of physical properties? thermal conductivity, electrical conductivity, physical state change, density, solubility, ductility, malleability, Answer: ALL ARE PHYSICAL PROPERTIES
- This is a good site to reveiw everything here
- Element hangman
- Kahoot quiz to practice chapter 3 topics
- Perhaps maybe an elements symbols quiz (a quiz will be worth 1/4 of a test)
- If time allows there are a lot of study links from Wednesday September 2 that we did not go over and you can do on your own.
- Study for the test on chapter 3. Friday we will review. Plan to take test early next week (maybe next Monday depending on how everyone is doing)
- CHAPTER 3 HANDOUT (10 PAGES ALL RELEVANT TO CHAPTER 3 BUT FROM ANOTHER TEXT CHAPTER 5)
- Do as much chapter 3 problems at the end of the chapter to help and make note of the ones that you had difficulties with so we can do them in class on Friday when we meet again (it will not be counted for your grade but it is suggested so you really know if you are getting the concepts right)
- Element quiz on Friday
- Again there are lots of links to go back and review like the 2nd one under today's agenda and a whole bunch on Wednesday September 2
Friday September 11
QUESTIONS:
AGENDA:
- What is the most abundant element in the universe?
- Top 3 elements found on earth?
- Top 3 elements found in human body?
- Are most elements found in nature in the elemental or in the combined (chemical compound) form? Name the the type of elements that are usually found in the elemental form?
- Oil and water do not mix. But they will if you were to add this to them. Why?
AGENDA:
- Collect homework 3.8 - 3.11 questions
- ELEMENT QUIZ
- By the way, Mrs. Frere does not use worksheets with this Chemistry book. Only uses the questions in the text!
- http://www.chagrinvalleysoapandsalve.com/idascorner/soap/how-does-soap-work
Monday September 14
QUESTIONS:
AGENDA:
HOMEWORK
- Write the formula for each of the following substances, listing the elements in the order given. a) A molecule containing two boron atoms and six hydrogen atoms b) A bolecule containing one carbon atom and four bromine atoms c) A molecule containing three hydrogen atoms, one phosphorous atom, and four oxygen atoms
- What particles make the greatest contribution to the mass of an atom?
- Which particles make the greatest contribution to the chemical properties of an atom?
- Are all atoms of the same element identical? if not, how can they differ?
- How many protons, electrons and neutrons are contained in the nucleus of each of the following atoms assuming they are uncharged? a) ₉₅²⁴¹ Am b) 133/55 Cs c) 56/25 Mn
- Which group do each of the following elements belong? a) iodine b) Ca c) Na d) lithium e) Kr f) sodium g) Ne
- List the gaseous elements that exist as diatomic molecules?
- List some properties of a substance that would lead you to believe it consists of nonionic compounds?
AGENDA:
- KEYWORDS and SUMMARIES are listed in your textbooks so I won't repeat them here.
- Go over the review packet which says chapter 5 on it (it is the same as chapter 3 for our textbook, covers the same material)
- See how the class responds to test being tomorrow. I can add 10 more questions to the last kahoot that I would play right before the test for review.
- The Nucleus: Crash course- open ed
- History of atomic theory - open ed
- setting the periodic table - game
- Types of chemical bonds
- Web elements- the periodic table on the www- game
- Carbon is a tramp
HOMEWORK
- Study for test.
- Do whatever homework you didn't do for review and also to get some partial credit.
Tuesday September 15
QUESTIONS:
- What element has the highest atomic weight?
- What type of gas did the Hindenburg use?
- For the following pairs of ions, use the concept that a chemical compound must have a net charge of zero to predict the formula of the simplest compound that the ions are most likely to form. a) Fe³⁺ and P³⁻ b) Fe³ and S²⁻ c) Fe³⁺ and Cl⁻ d) Mg²⁺ and Cl⁻
- All sites here can be seen by going to http://www.NeoK12.com and username is tsanelahachi and password is 0808jtr
- Label the diagrams of an atom
- Periodic Table - labelling activity
- Periodic table quiz (practice)
- Element Symbols practice quiz
- Atomic number and mass matching quiz
- atomic particles matching quiz 1
- atomic particles matching quiz 2
- Rutherford's gold foil experiment matching quiz
- Go over the review packet and then go over other "things you should know" for test
- KAHOOT.IT
- KEYWORDS and SUMMARIES are given in text so I will not repeat here
Wednesday September 16
TEST DAY
Friday September 18
OBJECTIVES:
QUESTIONS:
HOMEWORK:
- Learn to name binary compounds of a metal and nonmetal
- Learn how to name binary compounds containing only nonmetals
- Learn the names of common polyatomic ions ad how to use them in naming compounds
- Learn names for common acids and how the anion composition determines the acid's name
- Learn to write the formula of a compound, given its name
QUESTIONS:
- What do you think binary compounds are? Can you name some examples?
- How does heavy metal toxicity affect you? part 1 - 8:54 How does heavy metal toxicity affect you? - 4:44
- Mercury poisoning symptons and other heavy metals Part 1 - 6:06 Mercury poisoning symptoms and.. part 2 - 7:34 part 3- 3:41
HOMEWORK:
- READ SECTIONS 4.1 AND 4.2
Monday September 21
QUESTIONS:
AGENDA:
- In naming binary compounds the ____ is always named first and the ____ second.
- For Type I ionic compounds (the most basic) the anion is named by taking the first part of the element name and adding _____
- Name the following: KCl, ZnS, CaBr₂, BaH₂, Al2S₃, NaCl, KI, CaS, CsBr, MgO
- Name the following: SnI₄, K₃N,, HgO, RbF, NaH, CrF₂, MgBr₂. Li₂O
AGENDA:
- Try to do the homework in class and if done do the following links
- Ionic compound practice quiz
- Read section 4.3 and answer FOCUS QUESTIONS SECTIONS 4.1 - 4.3 (five questions)
- Ionic compound practice quiz (ODDS) for homework and we will do the ionic compounds worksheet to do tomorrow in class.
Tuesday September 22
QUESTIONS:
- Ionic compounds worksheet
- What is "hard" and "soft" water and what are the two most commom impurities in one of the forms?
- Alkaline earth metals are harder than it looks Test for hard water video-1:21 (having a high mineral content in your water) Hard water build up looks like soap scum
- Here are the transition metals that are Type II cations meaning they can have different charges. There is Cu⁺ and Cu²⁺ which is copper (I) and copper (II). There is Fe²⁺ and Fe³⁺ which is Iron (II) and Iron (III), and Co²⁺ and Co³⁺ which is cobalt (II) and cobalt (III). And there is Sn²⁺ and Sn⁴⁺ which is tin (II) and tin (IV), Mn²⁺ and Mn⁴⁺ which is manganese (II) and manganese (IV), and Pb²⁺ and Pb⁴⁺ which is lead (II) and lead (IV). Then there is Hg₂²⁺ and Hg²⁺ which is mercury (I) and mercury (II) but mercury (I) is a special case whose ions always occur bound together in pairs to form Hg₂²⁺
- Zinc, silver, and cadmium are transition metals that form only one ion (2+, +, and 2+ respectively)
- NH₃ is ammonia, NOT Nitrogen trihydride, H₂O is water, NOT dihydrogen oxide. Ammonia and water are always referred to by their common names.
- NO (Nitric Oxide) regulates blood pressure by dilating blood vessels. Good for sickle cell anemia.
- We are going to try something new. I want to make use of everything in the text so I am going to assign the chapter review questions from now on. And for the focus questions we will just go over them in class together. There are 51 chapter review questions for chapter 4 so start from question number one and since you were supposed to have read 4.3 then you can do up to #13 today. I am going to score these chapter reviews so that you are making sure to try and get the correct answers.
- Read section 4.4 and do up to #21 in chapter review.
Wednesday September 23
QUESTIONS:AGENDA;
Electrical Conductivity lab -
- Determining formulas for ionic compounds
- Acids and Bases practice assessment (probably will not get up to this point) Understanding acids and bases conceptually
- Chemical Nomenclature practice assessment (probably will not gety up to this point)
- Acids and bases worksheet (probably will not get here)
Electrical Conductivity lab -
- solution: (water, table sugar, sodium chloride, potassium nitrate, HCl, vinegar) - missing conductivity tester
- Introduction: Some compounds contain ions. We can test for this condition by dissolving a compound in water. If the compound contains ions, the ions will be dispersed throughout the water and free to move. The movement of the ions in a solution enables them to conduct a current.
- Notes: vinegar+ammonia (yes), HCl in water (yes), HCl (no)
- Again- read section 4.4 and do up to #21 in chapter review
Friday September 25
QUESTIONS:AGENDA:
- Chemistry of compounds:
- HCO₃⁻ is commonly called bicarbonate ion
- CN⁻ commonly known as the cyanide ion
- Ionic compounds: usually form crystals (solid), have high melting & bioling points, hard/brittle, usually soluble in water, almost never flammable
- Covalent compounds: are usually liquids or gases at room temp, lower mp & bp, non-soluble in water, almost always flammable
Monday September 28
QUESTIONS:
- From the pictures above what do you think the strong force is?
- The bi- prefix means: add H+ ions to the anion until its charge is -1. therefore you would have the following: Carbonate ion would be CO₃²⁻⁻ so the Bicarbonate ion is CHO₃⁻ Phosphate ion = PO₄³⁻ so biphosphate ion = PH₂O₄⁻ which would make sodium biphosphate = NaH₂PO₄
- http://antoine.frostburg.edu/chem/senese/101/index.shtml
- Gizmos
- End of chapter questions
Wednesday September 30
Sticky water and soapy water - 3:56
Friday October 2
QUESTIONS:
- Which is more healthy for you, hard water or soft water?
- Explain what a binary compound type I is
- Explain what a binary compound type II is
- When do you use the prefixes mono, bi, tri, tetra, penta, etc..
- Classzone Quiz A, B, and C
- Click on the nomenclature of binary nonmetal nonmetal compounds below
- quiz polyatomic ions from online chemistry
- classifying compounds quiz from online chemistry
- Naming ionic compounds problem sheet
- Naming molecular compounds problem sheet
- Writing formulas from names problem sheet
| nomenclature_of_binary_nonmetal_nonmetal_compounds.ppsx | |
| File Size: | 72 kb |
| File Type: | ppsx |
Monday October 5
AGENDA:
- If molecules were people - 3:24
- Naming Compounds - 10:51 or try Nomenclature Crash Course Chemistry #44 - 9:24 or How to Speak Chemistrian - 10:42
- brainpop: chemical bonds, ions, compounds and mixtures, chemical equations, atoms, moles, atomic model, body chemistry, nanotechnology, salt, measuring matter, diffusion, temperature, radioactivity, plastic, carbon dating, crystals, isotopes, matter changing states, metals, mineral identification, periodic table of elements, ph scale, property changes,
Tuesday October 6
AGENDA:
- SUBSTITUTE TEACHER TODAY AND TOMORROW, MR. ROSEN IS OUT WITH A COLD
- Use today and tomorrow to finish up all your chapter 6 packet and also all the chapter review questions (odds or evens) to be turned in next time we meet. And you should be ready to take the test.
- Classzone Quiz A, B, and C good to practice with these quizzes.
- You can go ahead and start reading the next chapter in the book if you think you are good with the current chapter.
FRIDAY OCTOBER 9
AGENDA:
- Review for test
- Try this nomenclature quizlet with 45 problems
- Classzone Quiz A, B, and C I suggest doing test C before going over it Monday. Start getting your end of chapter questions done as well.
Monday October 12
QUESTIONS:AGENDA:
- Another way to remember those polyatomic anions
- Rules for nomenclature here
Tuesday October 13
TEST ON NOMENCLATURE CHEMISTRY
HOMEWORK:
HOMEWORK:
- Start reading chapter 5: Measurements and Calculations sections 5.1 to 5.3 and do the section problems at end of 5.3 (Scientific Notation, Units, Measurements of length, volume and mass
- Also should do the chapter review problems (odd or even) up to #20.
Wednesday October 14
QUESTIONS:AGENDA:
- Collection of chemistry demonstrations 3:47 (liked artificial snow, lighting candle smoke, and ping pong) ice cream too.
- Conductivity of metals apparatus. The coefficients of heat transmission are as follows: (Aluminum 0.00203, steel 0.00062, copper 0.00404and brass 0.00142). Fill a shallow dish with about 1.5 inches of hot water. Stand the metal ends of the conductivity appratus in the hot water. See how the heat travels up the metal strips. Answer the following: 1) What is your prediction as to which metal will transmit the heat fastest? next fastest? next fastest? slowest? 2) Explain why you chose the order you did? 3) Did the outcome match your predictions?
- Describing a chemical reaction: In this activity you will produce a chemical reaction in which physical and chemical changes occur. You will write a description of your observations and translate it into a chemical equation using chemical notation. 1) Add 2.5 grams of copper chloride to a beaker containing 50mL of water. 2) dissolve with stirring stick 3) place piece of aluminum foil in solution and hold by stirring stick. 4) STORE PORTION OF SOLUTION IN PETRI DISH to observe precipitate (ppt) later. .. picture of precipitate below... WRITE DOWN 1) What you observe during reaction 2) write down element symbols that represent compound copper chloride 3) Write down statement that describes the reaction using reactants, arrow and description of reaction results (products) 4) Write down element symbols that represent white compound in the petri dish
Friday October 16
OBJECTIVES:
AGENDA; CHAPTER FIVE: MEASUREMENTS & CALCULATIONS
- Show how numbers can be expressed in scientific notation
- Learn the English, metric, and SI systems of measurement
- Use the metric system to measure length, volume and mass
- Learn how uncertainty in a measurement arises and express uncertainty using significant figures
- Learn to determine the number of significant figures in a calculates result
- Learn how dimensional analysis can be used to solve various types of problems
- Learn the three temperature scales and how to convert from one to another
- Define density and its units
AGENDA; CHAPTER FIVE: MEASUREMENTS & CALCULATIONS
- Go over Measurement and Calculations (Chapter 5) and let me know when you cannot understand something. Start working on chapter review questions (odds or evens up to problem #62) due next Friday 10/23
- Focus questions sections 5.4 - 5.5 due Tuesday 10/20
- Focus questions sections 5.6- 5.9 due Wednesday 10/21
- Chapter Review Five (all odds or evens up to #62) due Friday 10/23
- CHAPTER FIVE TEST FRIDAY 10/23
Monday October 19
QUESTIONS:
- How would you write the 93 million in scientific notation?
- Express 3.5 X 10⁻⁴ as a regular number
- Which is longer, a meter or a yard? A kilometer or a mile?
- List all the Power of 10 prefix names and symbols that you know from 10⁻ⁱ² to 10ⁱ² (10⁻ⁱ², 10⁻⁹, 10⁻⁶, ... is pico (p), nano (n), milli (m), etc... )
- How many inches in a foot? How many feet in a yard? which is longer, a meter or a yard?
- How many pints in a quart? How many quarts in a gallon? If one gallon is about 4 Liters then 1 Liter is about 1 _____.
- A unit for quantifying mass is also seen quantifying volume. What unit is this?
- The definition of the meter now depends on the definition of the _____.
- Table of unit conversion (cooking) compares common terms that you hear but not good to use when doing conversions later on
- Measurement Matrix (At the very least do the intro to metric system and the Intro to US standard units)
- An ounce is a measure of mass. A fluid ounce is a measure of volume.
- Know that 1 inch = 2.54 cm
- Know that 1 cups = 8 fluid ounces
- Know that 1 mL = 1 cc (cubic centimeter - cm³)
- Know that 1L = 1.06 qt = 1dL³
- Know that 1 lb. = 453.6 g = 16 oz.
- Know that 1 Kg = 2.205 lb
- Know that 12 inches = 1 ft.; 3 ft. = 1 yd (yard); 1 m = 1.094 yd; 1 mi (mile) = 5280 ft = 1760 yd
Tuesday October 20
QUESTIONS:
- There are 2 igals in 1 odonku, and 6 odonkus in 4 falgers. If you have 3 igals, how many falgers is this?
- At what temperature does water freeze and boil in the Fahrenheit scale? In the Celcius scale?
- What are the units used for both Fahrenheit and Celcius?
- What is called the absolute scale? What are the freezing and boiling temperatures for this scale? What are its units?
- Which temperature unit are the smallest?
- In a series of calculations, carry the extra digits through to the final result and THEN round off. Use your calculator until you arrive at the final number (the answer) and then round off.
- SIGNIFICANT FIGURE RULES HERE
- Students will work out dimensional analysis problems on the board
- ℃ + 273 = Temperature in Kelvins
- ℉ = 1.80 (℃) + 32; ℃ = (℉ - 32) / 180
|
|
| ||||||||||||||||||
Wednesday October 21
QUESTIONS:
- Which element has the highest density? The lowest?
- Isopropyl alcohol has a density of 0.785 g/mL. What volume should be measured to obtain 20.0 g of the liquid?
- The ratio of the density of a given liquid to the density of water at 4℃ is called what? What is its unit called? What measures it?
- RAINBOW DENSITY COLUMN LABORATORY - see page 142-143 in text
- Density RAINBOW performed - 2:23
- HANDOUT TO BE FINISHED AND TURNED IN BY END OF CLASS:
- Know the density for water is 1.00 Kg / L
- Review for test on Friday 10/23
Friday October 23
QUESTIONS:
- What is it called when all absolute motion stops?
- What temperature scale is based upon water?
- BRAINPOP: PRECISION AND ACCURACY, Metric Units, Metric vs Customary, Customary Units, Measuring Matter, Numbers and Operations, Rounding, Temperature, Scientific Notation,
- TEST: MEASUREMENTS AND CALCULATIONS
Monday October 26
OBJECTIVES:
AGENDA: CHAPTER SIX: CHEMICAL COMPOSITION
- Understand the mole concept and Avogadro's number and convert among moles, mass and number of atoms in a sample
- Understand the definition of molar mass
- Learn to convert between moles and mass of a given sample of a chemical compound
- Learn to find the mass percent of an element in a compound
- Understand the meaning of empirical formulas
- Learn to calculate the molecular formula of a compound, given its empirical formula and molar mass
AGENDA: CHAPTER SIX: CHEMICAL COMPOSITION
- Use individual boards, kleenex and markers
- 1 amu = 1.66 x 10⁻²⁴ g
- (atomic mass units) amu = Hydrogen = 1.008 , Carbon = 12.01, Nitrogen = 14.01, Oxygen = 16.00, Sodium - 22.99, Aluminum = 26.98
- 1 mol = 6.022 x 10²³ atoms = Avogadro's number.
- mol stands for mole and not molecule
- You don't need to memorize the average atomic masses of elements. These masses are given in the periodic table.
- A Mole is a Unit song - 3:25
Tuesday October 27
QUESTIONS:
- What does the average atomic mass of an element represent? What unit is used for average atomic mass?
- Express the atomic mass unit in grams. ?
- What does one mole of a substance represent on a macroscopic, mass basis?
- How do we know that 16.00 g of oxygen contains the same number of atoms as 12.01 g of carbon, and 22.99 g of sodium?
- How do we kow that 106.0 g of Na₂CO₃ contains the same number of carbon atoms as does 12.01 g of carbon, but three times as many oxygen atoms as in 16.00 g of oxygen (O), and twice as many sodium atoms as in 22.99 of sodium?
- Clozed statement using vocabulary here...
- Use individual boards, kleenex and markers
- Video- The mole and Avogadro's number - 5:28
- brainpop: The mole
- We just released Mole Day which was 10/23 starting at ending at 6:02
- 1 amu = 1.66 x 10⁻²⁴ g
- (atomic mass units) amu = Hydrogen = 1.008 , Carbon = 12.01, Nitrogen = 14.01, Oxygen = 16.00, Sodium - 22.99, Aluminum = 26.98
- 1 mol = 6.022 x 10²³ atoms = Avogadro's number.
- How many atoms of aluminum are in a mole of aluminum
Wednesday October 28
AGENDA:
- Many students non present as to the testing going on today. Therefore we will spend this block period catching up on make up work, make up tests, etc... with the remaining time reading and completing each new section and its focus questions as they come.
- If you get done with the chapter then there are always the chapter review problems due the same day as the test and some of you did not do this for the last one and it brought the A grade down to B.
- There will be a handout passed out which coincides with what we are studying now (Chemical quantities & composition- mole, amu, etc...) We are on chapter 6 in our book but the handout will say chapter 7 because it goes along with a different text.
- Perhaps the Instant Snow Polymer that I ordered: Add water to this granular white powder and watch it instantly expand to 40 times its original volume. (Simply add table salt to reverse the reaction- maybe, because this was for a slightly different product.) The result is a fluffy artificial snow that feels as cool as it looks! It won't "melt" but it can be dehydrated and re-used. Great for teaching endothermic and exothermic reactions or as part of a polymer lab. This is a great demonstration of interest to students and educators of ALL ages and abilities. 100 grams = 2 gallons of snow (might as well use the whole 100 grams but get it on video)
| student_ch_9_the_mole.ppt | |
| File Size: | 1136 kb |
| File Type: | ppt |
Friday October 30
AGENDA:
- Brainpop: The Mole - Make sure you read the FYI (For your information) and Q&A because you can be quizzed on them
- The Mole (ACTIVITIES will be printed out and if not turned in by end of class will be for homework- 2 pages)
Monday November 2
AGENDA:
- Watch Chemistry VCR video now that we have gotten to moles
- Start doing the practice problems in the book and also the Chapter reciew
Tuesday November 3
QUESTIONS:
- Calculate the number of moles of atoms and the number of atoms in a 25.0 g sample of calcium.
- Calculate the number of moles of atoms and the number of atoms in a 57.7 g sample of sulfur
- Calculate the number of atoms in a 23.6 mg sample of zinc.
- Calculate the number of atoms in a 128.3 mg sample of silver
- Does the value of Avogadro's number affect the relative masses of the atoms of the elements?
- Chromium (Cr) is a metal that is added to steel to improve its resistance to corrosion (for example, to make stainless steel). Calculate both the number of moles in a sample of chromium containing 5.00 x 10²⁰ atoms and the mass of the sample.
- How would you say the number mole in regular language? (hint: 6.022 x 10²³ = 602.2 x 10²¹)
- STUDENTS MUST REALIZE THAT AVOGADRO'S NUMBER OR ATOMIC MASSES ARE NOT EXACT. THEREFORE THEY CAN LIMIT THE NUMBER OF SIGNIFICANT FIGURES IN A RESULT.
- COMPLETE THE BRAINPOP ACTIVITY IF YOU HAVE NOT DONE SO ALREADY
- FOR GRADING- AND DUE TOMORROW - ALL OF THE ABOVE QUESTIONS AND ALSO THE BRAINPOP ACTIVITY (BOTH PAGES)
Wednesday November 4
QUESTIONS:
- We will go over the questions that were given yesterday as well as with the brainpop handout
- Methane is CH₄ so how many moles of Hydrogen does 1 mole of methane contain? How about carbon?
- What is the mass of one mole of diatomic oxygen gas?
- Aluminum = 26.98; Gold = 196.97; Iron =55.85; Sulfur = 32.07; Boron = 10.81; Xenon = 131.3 Use this information and your conversion factors to find the mass of a) 0.25 mol Al b) 0.500 mol Au c) 5.0 mol Fe d) 25 mol S
- What is the molar mass of water?
- calculate the mass of 1.48 mol of potassium oxide
- The Bean Lab: An Investigation of Moles (Handout) FYI one score = 20 and one ream = 500 and one gross = 144
Friday November 6
QUESTIONS:
- How many water molecules are in a 10.0-g sample of water?
- Sucrose, or table sugar, has the formula C₁₂H₂₂O₁₁ How many molecules of sugar are in a 5.00-lb bag of sugar?
- Turn in finished Lab from yesterday
- After going over questions read on your own chapter 6.5 to 6.8 doing all of the practice problems and chapter review questions that coincide with the section that you are reading.
Monday November 9
QUESTIONS:
- What is the molar mass of K₂O? 2 x (39.10) + 1 x (16.00) = 94.2 g / mol
- Determine the percent composition by mass for water.
- Handout CHEMICAL QUANTITIES (pg 63 - 70)
- Crash Course in Chemistry #6: Stoichiometry, the chemistry for massive creatures- 12:46
Tuesday November 10
QUESTIONS:
- Chapter 6 QuizA (from online companion)
- Students should work on the chapter 6 quizA questions above and, if done, complete their chapter review questions
Thursday November 12
QUESTIONS:
- The _____ formula gives the lowest whole-number ratio of moles of atoms in a mole of molecules.
- If a dozen large eggs weighs 0.75 kg and the net weight of eggs in a box is 6.0 kg, there is(are) _____ dozen eggs in the box.
- If a dozen small eggs weighs 0.30 kg and the net weight of eggs in a box is 2.4 kg, there are _____ egg(s) in the box.
- The masses on the periodic table for each element can be used to calculate the molar mass if they are used with units of amu.
- The molar mass of CaCl2 is _____.
- The atomic masses on the periodic table represent the average mass of an atom of that particular element? (true / false)
- To determine the molar mass of ionic substances, the mass of the electrons gained or lost must be taken into consideration. (true/false)
- A sample of silver with 18 x 10^21 atoms would have a mass of _____.
- A sample of methane (CH4) with 2.7 x 10^20 molecules would have a mass of ____ (A) 16 g (B) 7.2 x 10-3 amu (C) 16 amu (D) 3.6 x 104 g (E) 7.2 x 10-3 g10.
- A 9.5 g sample of magnesium chloride contains _____ chloride ion(s). (A) 2 (B) 0.2 (C) 1.2 x 1023 (D) 2.0 x 1023 (E)5.0 x 10-24
- 11. A 30 g sample of gold atoms is _____ mole(s). (A) 6.6 (B) 0.15 (C) 9.2 x 1022 (D) 9.2 (E) 5.9 x 103
- 12. A 200g sample of CaCO3 has _____ mole(s). (A) .5 (B) 2.9 (C) 2.00 (D) 0.500 (E) 213.
- Percent is always calculated as the part divided by the whole multiplied by 100%. (A) true (B) false
- Table sugar, sucrose (C12H22O11), has a molar mass of _____. (A) 342 g (B) 342 amu (C) 290 g (D) 180 g (E) 45 g
- There is (are) _____ mole(s) of diatomic iodine molecules in 19.9 g of the solid. (A) 254 (B) 0.0783 (C) 0.157 (D) 0.0392 (E) 12.8
- There are _____ moles of oxygen atoms in 2 moles of Al2(SO4)2. (A) 18 (B) 4 (C) 3 (D) 12 (E) 16
- The mass percent of carbon in Na2CO3 is _____. (A) 16.2% (B) 88.7% (C) 11.3% (D) 23.5% (E) 43.3%
- The empirical formula of the compound containing 42.3% chromium and 57.7% chlorine by mass is _____.(A) CrCl₄ (B) CrCl (C) CrCl₂ (D) CrCl₃ (E) Cr₂Cl₂
- The mass percent of oxygen in CO2(SO4)2 is _____. (A) 11.8 (B) 41.3% (C) 3.90% (D) 47.3% (E) 37.2%
- When given only mass percent data, the molecular and structural formulas can be determined. (True or false)
- No computers and seat change
- 20 questions above to be worked on today and then due by end of period tomorrow Friday November 13
Friday November 13
Agenda:
- complete the 20 questions in class and turn in by end of class
- if finished with 20 questions then work on chapater review questions
- I will be doing a couple problems up on the board and if you have any specific questions now is the time to ask
- USING CHEMISTRY TO UNDERSTAND LOST WORLDS: You should know that a high phosphorous content in a given soil is indicative of animals being stored in that area because animal dung has a high phosphorous content. If there is also a high calcium content then that is indicative of being upon a slaughter house where animals were butchered since there is a high amount of calcium in bones and teeth. There is also a high phosphorous amount in animal flesh. On the other hand areas with depletion of phosphorous, calcium, magnesium, nitrogen and sulfur are indicative of areas used for gardening.
Monday November 16
QUESTIONS:
- #19 from yesterday
- Continue with Friday's problems and then start finishing chapter review. Any questions I am here to be asked. You have today and tomorrow for review for test on Thursday.
- TEST ON THURSDAY
Tuesday November 17
QUESTIONS:
1 How many moles of nitrogen atoms are contained in one mole of Ba(NO3)2?
2 Mole is to atom as gram is to — A amu B mass C molecule D particle
3 What is the total number of atoms contained in 2.00 moles of helium?
A 15.999 B 32.0 C 6.02 1023 D 1.20 1024
4 A compound has the formula MgSO4•7H2O. Its chemical name is --
A aqueous magnesium sulfate B magnesium sulfate pentahydrate C magnesium sulfate heptahydrate D magnesium sulfate decahydrate
A1 B2 C6 D9
5 The molecular formula of a compound is X6Y3. What is the empirical formula for this compound?
A X6Y B XY3 C X2Y D XY2
6 Zinc is used as a coating on iron and steel to prevent corrosion. What is the mass, in grams, of 0.0650 mol Zn? A 3.25 g B 3.90 g C 3.94 g D 4.25 g
7 Indium (In) is a relatively rare element that never occurs as a free metal. It isusually found in a compound that contains 70.48% In and 29.52% S. What is the empirical formula for this compound? A InS B In2S3 C In3S5 D In6S9
8 A student measures 10.0 g of hydrated sodium carbonate (Na2CO3•xH2O) and places it in a crucible. After heating, 3.7 g of anhydrous sodium carbonate (Na2CO3) remains. What is the formula for the hydrate? A Na2CO3•2H2O B Na2CO3•5H2O C Na2CO3•8H2O D Na2CO3•10H2O
9 Potassium nitrate, also known as saltpeter, is used in matches. What is the percent by mass of potassium (K) in potassium nitrate (KNO3)?
A 38.67% B 45.94% C 55.71% D 56.58 %
10 Baking soda is the common name for sodium hydrogen carbonate (NaHCO3). What is the mass in grams of 2.75 moles of sodium hydrogen carbonate? A 63.2 g B 84 g C 210 g D 231 g
11 A mole of 12 C atoms will have a total mass of — 6
A 12 kg B 12 g C 12amu D 6amu
AGENDA:
- Chemistry lab manual glencoe.pdf Chapter 11.1 (page 81) xtra practice
- Chemistry reviewing student edition.pdf Stoichiometry (pages 23 and 24) xtra practice
- PVDF is a plastic material that is piezoelectric (when it is physically deformed it produces an electrical current or vice versa) and pyroelectric (develops electric potential in response to temperature change). Used for , example, microphone.
- The mass of the electron is so small that Na⁺ and Na have the same mass for our purposes even though Na⁺ has one electron less than Na. Also, the mass of Cl virtually equals the mass of Cl⁻ een thogh it has one more electron than Cl
- William Perkin- while tyring to synthesize artificial quinine he accidentally made a purple dye which he called mauve. Before this purple was color of the wealthy where it took 9000 mollusks to make 1 g of purple dye. He then went on to develop another dye he called alizarin which was red. He sold his factory and made enough money to retire at age 36.
- The major use of ammonia is as a fertilizer to furnish nitrogen atoms to growing plants.
|
| ||||||||||||
Thursday November 19
AGENDA:
- Review for test
- CHAPTER 6 TEST
- LABORATORY: DECOMPOSING COPPER OXIDE
Monday November 30
OBJECTIVES:
- Learn the signals that show a chemical reaction has occurred
- Learn to identify the characteristics of a chemical reaction and the information given by a chemical reaction
- Learn to write a balanced equation for a chemical reaction
- Name 4 clues that a chemical reaction has occured. 1) The _____ changes. 2) A ______ forms. 3) _____ form and 4) _______ is produced.
- Are the above 4 all proof that a chemical change has taken place? Whichi ones are not and give example?
- Chemistry games: elements, polyanions, ionic vs molecular bonds, weak/strong/nonacids vs weak/strong/nonbases, etc...
- Make up test - Tuesday
- Classification of chemical reactions problems
- Prediction products of chemical compounds problem sheet
Tuesday December 1
AGENDA:
- MAKE UP TEST
- READ CHAPTER SEVEN AND START DOING REVIEW QUESTIONS
- How to write balanced equations 1) write the appropriate reactants, products, and their states (solid, liquid, or gas) 2) Start writing coefficients with the most complicated molecule. Then proceed element by element to determine what coefficients are necessary so that the same number of each type of atom appears on both sides. 3) Check and make sure that the coeficients used give the same number of each type of atom on both sides of the arrow. Also check that the coefficients are the smallest integers that give a balanced equation.
Thursday December 2
QUESTIONS;
- The most common modern pain killer is ______ and its active ingredient is salicylic acid which is found in willow bark and converted in boiling water. It works by interfering with the production of prostaglandins, which are molecules that generate pain signals and cause inflammation. It also works to prevent clotting by _____ the blood.
- Besides masking pain and preventing inflammation, this drug appears beneficial in preventing ____ ______ and cataracts. However it could also cause bleeding in the digestive tract.
- practice balancing equations - do chapter reviews
Monday December 6
AGENDA:
- REVIEW HANDOUT PASSED OUT TO STUDY FOR FINALS (CHAPTERS 1 TO 7)
- Now is the time to ask any questions or ask me to show solutions to any problems.
SECOND SEMESTER
Monday January 4
SOLUBILITY RULES
OBJECTIVES
HOMEWORK:
- Learn some factors that cause reactions to occur
- Learn to identify the solid that forms in a precipitation reaction
- Learn to write molecular, complete ionic, and net ionic equations
- Learn the key characteristics of the reactions between strong acids and strong bases
- Understand electron transfer as a driving force for a chemical reaction
- Learn various classification schemes for reactions
- Dissolution of a solid in a liquid 1:12 (see how water being polar can dissolve a solid)
- Remember the following after a formula: (s), (l), (g), and (aq) and (aq) stands for aqueous which means ions dissolved in water.
- K₂CrO₄ (aq) + Ba(NO₃)₂ (aq) ► ppt = precipitate = solid
- Know that there will be K⁺, CrO₄⁻, Ba²⁺, and NO₃⁻ ions, all (aq) which two will combine to make the precipitate (ppt).
HOMEWORK:
- Read 8.1 and 8.2 and start trying to remember the general rules for solubility of salts (ionic compounds) in water (pg. 218)
- Review polyanions because we will be using those alot
Tuesday January 5
AGENDA:
- Practice solubility rules with examples given in the book: All nitrates are soluble, etc...
Thursday January 7
AGENDA: MAKING ROCK CANDY AND FACTS ABOUT CRYSTALS
- Solids come are either 1) amorphous, 2) without form, or 3) crystalline. Crystalline solids are definite, rigid shapes with clearly defined faces and come in 7 forms.
- A crystal is any solid material whose atoms are arranged in a definite geometric pattern.
- All crystals can be classified into one of seven basic shapes. They usually have flat planes that are set at fixed angles to one another.
- Some crystals are very rare and sell for high prices. Gemstones such as diamonds, emeralds, rubies, and sapphires are crystals
- Crystals are used in electronics, lasers, communications, barometers, pressure gauges and watches, to name a few
- Crystals grow then they come into contact with the same molecules or atoms they are made of.
- They are inanimate which means they are not alive.
- To grow crystals in sugar-water the water must be saturated (dissolved all the sugar it possibly can). The warmer the water, the more sugar the water will dissolve before it becomes saturated.
- When a sugar solution is cooled, it becomes super-saturated (more sugar in it than can stay in liquid form) The sugar then begins to crystallize (change from liquid to solid).
- Energy is released from the bond that is formed which attracts more atoms of sugar molecules in the solution.
- When done after seven days you will have made rock candy just like candy makers did more than one hundred years ago.
- Measure exactly 3/4 cup water and pour into one-quart saucepan. heat in microwave until boiling.
- Immediately pour bag of sucrose (or table sugar) into boiling water and begin stirring with a spatula
- Stir solution continuously with spatular on medium-high heat. the solution will appear milky or cloudy. after two minutes of heating and stirring, the solution will become clear so you can see the bottom of the saucepan. continue heating and stirring for about one more minute so that most of the grains of sucrose in the solution are completely dissolved (dont' get solution too hot or it will not grow crystals). As soon as all granules have dissolved, immediately remove from heat.
- Remove saucepan from heat and pour solution into the growing vessel. Put vessel on a stable heat resistant surface.
- insert bare end of the seeded stick through the hole in the bottom side of the lid. The coating on the stick is what the rock candy crystals will grow on. We refer to these as "seeds"
- Lower the seeded stick into the sugar solution in the growing vessel. Position the stick so that it is hanging in the center of the vessel.
- PRECIPITATION METHOD: Keep the growing vessel in a place that stays warm at a constant temperature somewhere between 70 and 85 ℉. Avoid shaking or disturbing the crystals as they are growing. Each day there will be notieceable crystal growth on the stick and on the bottom and sides of the vessel (about 7 days). When the crystals stop growing, the sugar solution is in equilibrium The water is no longer supersaturated, but saturated.
- EVAPORATION METHOD (optional): If you want to make the crystals grow even larger punch or cut holes in the plastic lid of the growing vessel. The holes will allow air into the vessel and will cause the water in the sugar solution to evaporate. As water in the solution is removed by evaporation, more sugar must change from liquid to solid. The crystals will grow much slower but they will keep growing until all the water has evaporated.
Monday January 11
AGENDA:
- SEE WHAT YOU KNOW WORKSHEET: Classifying Elements: terms review, Making chemical formulas with chemical names, working with chemical formulas, physical changes
- Know your polyatomic ions in table 4.4, page 100. We will have a quiz tomorrow.
- review the driving forces we have learned so far: 1) the formation of solid (this is where a precipitate forms). The solid that forms is called a precipitate, and the reaction is known as a precipitation reaction. You need to know solubility rules with this reaction which are as follows:
- Most nitrates (NO₃⁻) salts are soluble.
- Most salts of Na⁺, K⁺, and NH⁺ are soluble.
- Most chloride salts are soluble. Exceptions are AgCl, PbCl₂ and Hg₂Cl₂
- Most sulfate salts (SO₄²⁻) are soluble. Exceptions are BaSO₄, PbSO₄, and CaSO₄
- Most hydroxide compounds are not soluble. Exceptions are NaOH and KOH. However BaOH₂ and CaOH₂ are moderately soluble
- Most sulfides (S²⁻_, carbonates (CO₃²⁻), and phosphates (PO₄³⁻) are not soluble.
- *when I say "not soluble" in reality there is a very tiny amount that dissolves but not possible to detect from naked eye
Tuesday January 12
AGENDA:
- Driving forces (precipitation and water)
- Review of ppt (precipitation reactions) using solubility rules (did all section 8.1 and section 8.2 problems on board)
- Intro to acid/bases (forms water) acids produce H⁺ and bases produce OH⁻
- Describing reactions in aqueous solutions: molecular, complete ionic, and net ionic equations
- spectator ions are ions that are present in the solution both before and after the reaction and therefore do not participate directly.
- strong acids and bases can split up into electrolytes (ions) easily (100%)
- Reminded that there will be a quiz on the polyanions (page 100 table 4.4) tomorrow
- Assigned homework problems individually to students: Ella (1, 5, 8-10(a)), Alana (2,6, 8-10(b), Gemma (3,7, 8-10(c), Taylor (3, 8-10(d))
Thursday January 14
AGENDA:
- Answer homework problems (if you did it you will get 100% for homework grade today)
- Molecular view of solution formation
- Brainpop: compounds & mixtures; (vocabulary sheet homework)
- Brainpop: chemical equations (vocabulary sheet homework)
- QUIZ TOMORROW - POLYATOMIC ANIONS PAGE 100 TABLE 4.4
- Assigned homework: Alana (11a,d, 12a, 13a), Gemma (11b,e, 12b, 13b), Taylor (11c,f, 12c, 13c)
- (s), (l), (g), plasma game
- 2 sheets of vocabulary
- chapter review problems 14-16
- read 8.4 and be prepared to answer questions 17 to 23 in class tomorrow
Friday January 15
|
electrolyte
molecular water spectator ion |
hydronium or H⁺
dissolving with water acid-base molecular formula |
Hydrochloric acid
hydroxide or OH⁻ salt |
complete ionic
strong electrolyte weak electrolyte non electrolyte |
QUESTIONS:
- A strong _____is a substance that completely breaks apart into ions when dissolved in water. Thd resulting solution readily conducts an electric current
- In a _____ _____ equation all substances that are strong electrolytes are represented as ions.
- The ____ equation includes only those components that undergo a change in the reaction.
- The complete formulas for all reactants and products are called the _____ equation for the reaction.
- Ions which do not participate directly in a reaction in solution are called _____ ions and are not included in this type of equation?
- The Arrhenius definition of an acid : a substance that [produces _____ ions in aqueous solution.
- Putting H₂O over an arrow in an equation means what?
- HCl is called _____ when in aqueous solution. It is a _____ electrolyte
- What are the 4 strong acids that are named in the textbook?
- Aqueous solutions that exhibit basic behavior always contain ____ ions.
- What are the two strong bases mentioned in the book?
- When strong acids and strong bases are mixed ____ is formed. The other product is always an ionic compound called a _____, which remains dissolved in the water. This salt can be obtained as a solid by evaporating the water. The reactions of H⁺ and OH⁻ is often called an _____ reaction.
Tuesday January 19
AGENDA:
- Worked homework problems on the board
- Field trip permission slips given out
- Read Section 8.5 (oxidation-reduction aka "redox" reactions)
Thursday January 21
LOS ANGELES ZOO FIELD TRIP
Friday January 22
AGENDA:
- Oxidation-Reduction reactions involve a transfer of electrons
- Batteries, corrosion, and combustion are all oxidation-reduction reactions
Monday January 25
|
net ionic
combination water combustion melatonin Calcium carbonate acid-base |
spectator ions
molecular decomposition propane vitamin E transfer of electrons precipitation |
Hydrogen ions
solubility heat gasolinesingle-displacement free radicals double-displacement carbon dioxide |
hydroxide ions
complete ionic anti-oxidants oxygen oxidizing coal carbon |
QUESTIONS:
AGENDA:
- Stalactites and stalagmites in Carlsbad Caverns, New Mexico are formed from what? (Same as eggshells, limestone, marble, seashells, and coral)
- What is the driving force of an oxidation-reduction reaction?
- When a metal reacts with a nonmetal, an ionic compound is formed when the metal transfers one or more electrons to the nonmetal, the metal atom becoming a cation and the nonmetal atom becoming an anion. Therefore they can always be assumed to be ______ reactions.
- Two nonmetals can also undergo an oxidation-reduction reaction. We recognize these cases by looking for _____ as a reactant or product.
- The formation of a solid indicates a/an _____ reaction
- The formation of water indicates a/an _____ reaction
- The transfer of electrons indicates a/an _____ reaction
- When the two cations switch with the two anions this can be called a/an _____ reaction
- When an anion switches places between two different cations this is called a/an _____ reaction
- The formation of a gas can indicate a/an _____ reaction if it involves an H⁺ or OH⁻ forming water as another product.
- The _____ molecule and other _____ substances in the body can extract single electrons from the large molecules that make up cell membranes (walls), thus causing them to become very reactive. If enough of these changes accumulate, the body's immune system comes to view the changed cell as foreign and destroys it. In houseflies the accumulated damage from oxidation is linked to both the fly's vitality and its life expectancy. One study showed that flies that were forced to be sedentary showed much less damage from oxidation because of their lower oxygen consumption and lived twice as long as flies that had normal activities. _____ has been shown to protect against oxidation. Two substances known to be antioxidants are _____ and _____. Oxidation forms _____ (a species with unpaired electrons).
- Good evidence indicates that _____ contains one or more chemicals that protect against cancer by blocking tumor formation. One such chemical that has been identified is called sulforaphane.
- Reactions that involve oxygen and produce energy (heat) so rapidly that a flame results are called _____ reactions. They are, in fact, a special form of _____ reactions.
- The combustion of _____ is used to heat some rural homes. The combustion of _____ is used to power cars and trucks. The combustion of _____ is used to generate electricity.
- When a given compound is formed from simpler materials, we cal this a synthesis (or _____) reaction. It is a subclass of _____ reactions
- The reaction of hydrogen gas with oxygen gas to produce water, then, can be classified 3 ways: as an _____ reaction, as a _____ reaction, and as a _____ reaction
- When a compound is broken down into simpler compounds or elements they are called _____ reactions. They are usually accomplished by _____ or by the application of an electric current. They are also a subclass of _____ reactions.
- Clues that a combustion reaction is occurring is the presence of _____ and a _____ containing molecule as reactants. The products of many combustion reactions are _____ and ____.
- General rules on _____ help predict whether a solid - and what solid - will form when two solutions are mixed.
- 3 types of equations are used to describe reactions in solution. The ____ equation, the ____ equation and the ____ equation.
- _____ are not included in the net ionic equation.
- A strong acid is a compound in which virtually every molecule dissociates in water to give an _____ ion. Similarly, a strong base gives off _____ and cations in water.
AGENDA:
- Zn + I₂ ► ZnI
- 2Al(s) + 3I₂(s) ► 2AlI₃
- Thermite reaction: 2Al(s) + Fe₂O₃(s) ► 2Fe(s) + Al₂O₃(s)
- TEST WILL BE THURSDAY, FEBRUARY 4
- Know the 5 types of reactions:
- Precipitation reaction (a solid forms)
- Acid-base (acid + base makes water)
- Oxidation-Reduction reactions involve the transfer of electrons and consist of 3 different subsets which are (synthesis, decomposition, and combustion reactions) Synthesis (in most basic explanation) has 2 reactants and 1 product. Decomposition has 1 reactant breaking down into 2 products (opposite of synthesis) and Combustion uses O₂ as a reactant with an organic compound (CᵪHᵪOᵪ) usually forming CO₂ and H₂O
Tuesday January 26
AGENDA:
- Answer the fill-ins from yesterday 1-22
- Know the 5 types of reactions (look at board)
- Re-take of polyanion test
- Reminder that (aq) means aqueous which means that it is a solution of ions in water and that is soluble.
- If something is not soluble then it cannot be dissolved in water and remains or precipitates into a solid
- If something is oxidized then its charge has went up. For example when Na(s) [charge=0] goes up to Na⁺ [charge=1]
- If something is reduced then its charge has went down. For example when O₂ (charge 0) goes down to O²⁻ (-2 charge)
- In a oxidation-reduction reaction there is always something that gets oxidized and something that gets reduced. This means that there is a transfer of electrons. So the "driving force" of oxidation-reduction reactions are the transfer of electrons.
- e.g. 2Al(s) + Fe₂O₃(s) ► 2Fe(s) + Al₂O₃(s) Al (s) ►Al³⁺ + 3e⁻ (Aluminum goes from 0 to 3 charge so it is oxidized) and this means that it is a reducing agent in that it enables Fe (iron) to get reduced by giving it its electrons. Fe³⁺ + 3e⁻ ► Fe (s) (Iron goes from +3 to 0 so it is reduced) And this makes Fe(s) the oxidizing agent as it helps Al get oxidized by accepting its electrons.
- An oxidizing agent accepts electrons
- A reducing agent donates electrons
- Real life application: It is thought that the more you eat and use oxygen the more you age and the more your cell membranes can become oxidized and the more this makes the cell reactive. After too much activity the cell will be viewed as "foreign" and destroyed. Antioxidants are sold to help negate this (Vitamin E is an antioxidant). Basically the less oxygen you consume the less you age according to housefly studies. The more sedentary houseflies lived twice as long as the normal houseflies.
Thursday January 28
AGENDA (FOR THURSDAY AND FRIDAY):
- Go through the following links and have students take turns answering them.
- Quiz A
- Quiz B
- Quiz C
- Chapter 8: Dissolution of a Solid in a Liquid
- Chapter 8: Electrolyte Behavior
- Chapter 8: Precipitation Reactions
- Chapter 8: Proton Transfer
- Chapter 8: Neutralization of a Strong Acid by a Strong Base
- Chemistry games:
- Chapter 8: Electrolytes
- Chapter 8: Solubility Rules
Monday February 1
- SUBSTITUTE TEACHER: MR. ROSEN OUT SICK
- Visualization of solubility rules - you should be able to tell what will happen before the solutions are mixed
- Students should know all answers to the following quizzes
- Quiz A (by end of Monday)
- Quiz B (by end of Tuesday)
- Read entire chapter 8 (this should already have been done)
- Start doing review chapter questions (odds or evens, for questions with a-d answers pick two of them) due end of Tuesday
- TEST WILL BE FRIDAY SO WE WILL ONLY HAVE WEDNESDAY AND THURSDAY TO STUDY!! WE HAVE STAYED TOO LONG ON THIS CHAPTER
- MONDAY AND TUESDAY ARE YOUR DAYS TO STUDY AND DO ALL CHAPTER REVIEW QUESTIONS, IF DONE, THEN DO MORE (IF ODDS THEN DO EVENS, I WILL GIVE EXTRA POINTS FOR THIS)
- You should never have "nothing" to do. If done with everything play the chemistry "games" Chemistry games: that will help you memorize main concepts such as "oxidation", "polyatomic anions", "electrolytes", "molecular or ionic", "acids & bases 1", "acids & bases 2", "metric prefixes", "solubility", and "elemental symbols" - all concepts that you now know!!! ( 5 just from this chapter) - JUST GO TO "GAME" AND SELECT THE DIFFERENT GAME THAT YOU WANT TO PLAY AND THEN CLICK ON THE "NEW GAME" ALL THE WAY TO THE LEFT.
Tuesday February 2
AGENDA:
- Try these videos to help you get the concepts better:
- water and solutions - crash course chemistry #7 - 13:33
- acid base chemistry in solution - crash course chemistry #8 - 13:33
- Precipitation reactions- crash course chemistry #9 - 11:20
- Redox reactions - crash course chemistry #10 - 11:12
- I have included the studyguide below in case it helps any of you. Just download...
| survivingchemistryguidedstudybookhschemistrywithnysregentsfree.pdf | |
| File Size: | 5895 kb |
| File Type: | |
Tuesday February 2
AGENDA:
- KAhoot after watching animations and short videos (QUIZ)
- Tuesday, Thursday and Friday we will study for the test by keep going over new problems, each student taking turns answering them.
- Chapter review problems (odd or even questions up to #39)
- study for TEST NEXT MONDAY
Monday February 8
AGENDA:
- Addendum: A double displacement reaction is when two solutions of ionic solutes are mixed and the positive ions of the two solutes exchange anions. You know this as a precipitation reaction Example: AgNO₃(aq)+HCl(aq)►AgCl(s) + HNO₃(aq)
- Addendum: A singled displacement reaction is when a new element replaces a less active element in the compound. You know this as a Oxidation-Reduction aka Redox reaction. Example would be: Mg(s) +2HCl(aq) ► MgCl₂(aq)+H₂(g)
- A transfer of electrons from atoms of a metal (Na) to atoms of a nonmetal (Cl) is favored because metals tend to lose electrons and nonmetals tend to gain electrons. This would be a redox reaction
- What reaction(s) could the following be classified as? C(s) + O₂(g) ► CO₂(g)
- TEST TODAY!!!
- Start reading chapter 9
Tuesday February 9
AGENDA:
- TEST HAS BEEN POSTPONED DUE TOO MANY STUDENT ABSENCES
- The people that were here took a practice test and I do not feel that they know enough to pass the test with anything more than a "C" so I think one more day of going through practice problems will help. I see some areas that we really did not cover that much.
- "Salt" is a term in chemistry that basically means an "ionic compound". An ionic compound basically is a metal/non-metal interaction.
- NaCl is only "table salt" . There are many other salts.
Thursday February 11
AGENDA:
- Review for test
- TEST CHAPTER 8
Friday February 12
AGENDA: SECTION 9.1
- Intro chapter 9
- Sample sandwich (Balancing equation)
- the dozen (refresh your memory of the mole)
- The coefficients in a balanced equation represent the mole ratios. Example 2NaCl where 2 is the coefficient.
- The subscript would be represented by the 2 in CO₂. The superscript would be represented by the 3 in x³
- 1 mole = 6.022 x 10²³ units
- THE NUTS AND BOLTS OF CHEMISTRY: An average mass of a bolt is 10.64g and an average mass of nut is 4.35g. Suppose you are given about 1500g of bolts and about 1500g of nuts. Answer the following questions:
- How many bolts are in about 1500g? How many nuts are in 1500g?
- Which reactant is limiting? why is there a limiting reactant given that you have equal masses of each reactant?
- the limiting reactant was the one that had (fewer/more) pieces.
- What is the largest possible mass of product? how many products could you make?
- what is the mass of the leftover reactant? HAVE A GOOD 4 DAY WEEKEND
- Read section 9.2
- Have a good 4 day weekend.
Wednesday February 17
OBJECTIVES:
- Learn to use a balanced equation to determine relationships between moles of reactants and moles of products
- Learn to relate masses of reactants and products in a chemical reaction
- Carry out mass calculations that involve scientific notation
- Understand the concept of limiting reactants
- Learn to use the limiting reactant to do soichiometric calculations
- We died the NUTS AND BOLTS OF CHEMISTRY question (from last class) our last class. This is on page 254 at the top. Now you can start through it all over again and follow the directions and answer the questions 1 through 8 (a through d) and turn it in by the end of class and that will be counted as participation score. Also you need to start reading 9.2.
- You can do problems 1-4 in the chapter review and then do as much from 5-10 after you finish reading 9.2 I cannot stress how important it is to do all the questions that you can.
Thursday February 18
AGENDA:
- CH₄(g) +2O₂(g)►CO₂(g)+2H₂O(g)
- Can someone read off to me the above equation.
- What kind of equation is this?
- Is it more than one kind of equation?
- What is the driving force?
- Instead of saying "equals" for the arrow sign there is a better word for this, what is it?
- How many moles of carbon dioxide results from using one mole of methane? How about water vapor? (how much will form)? vapor
- How many moles of carbon dioxide results from using two moles of methane? How about water vapor? (use dimensional analysis to derive the mole ratio)
- With 2 moles of methane and 2 moles of oxygen gas what is the limiting reagent?
- How many moles of methane and oxygen would you need to make 8 moles of water vapor?
- What number of moles of oxygen gas is required to react with 7.4 moles of methane?
- How many moles of carbon dioxide gas will be produced by reacting 2.6 mol of oxygen with excess methane?
- What was the limiting reagent for number 11?
- Ammonia (NH₃) is used in huge quantities as a fertilizer. It is manufactured by combining nitrogen and hydrogen.
- Someone translate into an unbalanced equation.
- Balance the equation
- What kind of reaction is this?
- What is its driving force?
- What is the formula for the ammonium ion?
- Calculate the number of oles of NH₃ that can be made from 1.30 mol H₂(g) reacting with excess N₂(g).
- Hydrogen sulfide gas reacts with oxygen to produce sulfur dioxide gas and water according to the balanced equation 2H₂S(g) + 3O₂(g) ► 2SO₂(g) + 2H₂O(g)
- What kind of reaction is this? What is its driving force?
- How many moles of oxygen gas are required to react with 5.6 mol of hydrogen sulfide?
- How many moles of sulfur dioxide gas will be produced by reacting 7.3 mol of hydrogen sulfide with excess oxygen?
- How many moles of sulfur dioxide gas will be produced by reacting 7.3 mol of oxygen with excess hydrogen sulfide?
- Iron (II) oxide reacts with oxygen to form iron (III) oxide according to the balanced equation 4FeO(s) + O₂(g) ► 2Fe₂O₃(s)
- Which iron compound is the iron(II) oxide solid and which is the iron(III) oxide solid?
- What type of reaction is going on here and what is its driving force?
- What number of moles of oxygen gas is required to react with 2.4 mol of iron (II) oxide?
- What number of moles of iron (III) oxide solid will be produced by reacting 9.2 mol of iron (II) oxide with excess oxygen?
- What number of moles of iron (III) oxide will be produced by reacting 9.2 mol of oxygen with excess iron(II) oxide?
- Do the focus question (all five) from sections 9.1 - 9.2 and turn in before class
- READ SECTION 9.3 - 9.5 BEFORE COMING TO CLASS TOMORROW (they are all review of what we have already done, 9.3 about converting weight of something to moles and 9.4 about including scientific notation and 9.5 is just comparing equations.
Friday February 19
AGENDA:
- Steps for calculating the masses of reactants and products in chemical reactionsSTEP 6: Convert from moles back to mass
- STEP 1: Balance the equation for the reaction
- STEP 2: Write down whatever information is given first to start with (LOOK FOR NUMBERS!)
- STEP 3: Convert the masses of reactants or products to moles (USE PARENTHESIS - FIND MOLAR MASS - PUT SAME UNIT ON BOTTOM AS PREVIOUS UNIT (FROM STEP 2)
- STEP 4: Use the balanced equation to set up the appropriate mole ratio(s) (USE PARENTHESIS- USE COEFFICIENTS ROM BALANCED EQUATION- SAME UNIT ON BOTTOM/DENOMINATOR AS PREVIOUS NUMERATOR)
- STEP 5: Use the mole ratio(s) to calculate the number of moles of the desired reactant or product.
- STEP 6: Convert from moles to mass (grams)
- Five of the world's most dangerous chemicals - 10:44
- What mass of oxygen will be required to react exactly with 44.1 g of propane?
- C₃H₈ (g) + 5O₂(g) ► 3CO₂(g) + 4H₂O(g)
- What mass of carbon dioxide is produced when 44.1g of propane reacts with sufficient oxygen?
- Calculate the mass of water formed by the complete reaction of 44.1g of propane with oxygen.
- 2Al(s) + 3I₂(s) ► 2AlI₃(s).... Calculate the mass of I₂(s) needed to just react with 35.0g Al(s)
- Calculate the mass of AlI₃(s) formed by the reaction of 35.0g Al(s) with 495g I₂(s)
- DO FOCUS QUESTIONS ON PAGE 268 (4 QUESTIONS) SECTIONS 9.3 TO 9.5 - WILL COUNT FOR HOMEWORK POINTS. IT IS IMPORTANT TO START DOING HOMEWORK AS BY NOT DOING SO WE HAVE GOTTEN BEHIND SO NOW YOU WILL FIND THE PACE BEING FASTER AND AS A RESULT THERE WILL BE MORE HOMEWORK.
Monday February 22
AGENDA: Section 9.3
- Steps for calculating the masses of reactants and products in chemical reactions
- STEP 1: Balance the equation for the reaction
- STEP 2: Write down whatever information is given first to start with (LOOK FOR NUMBERS!)
- STEP 3: Convert the masses of reactants or products to moles (USE PARENTHESIS - FIND MOLAR MASS - PUT SAME UNIT ON BOTTOM AS PREVIOUS UNIT (FROM STEP 2)
- STEP 4: Use the balanced equation to set up the appropriate mole ratio(s) (USE PARENTHESIS- USE COEFFICIENTS ROM BALANCED EQUATION- SAME UNIT ON BOTTOM/DENOMINATOR AS PREVIOUS NUMERATOR)
- STEP 5: Use the mole ratio(s) to calculate the number of moles of the desired reactant or product.
- STEP 6: Convert from moles back to mass
- Calculate the mass of carbon dioxide gas that is required to react with a solution containing 10.0g of sodium hydroxide using the following formula: 2NaOH(aq) +CO₂(g) ► Na₂CO₃ + H₂O(l)
- Calculate the mass of sodium carbonate that is produced when 10.0g of sodium hydroxide reacts with an excess of carbon dioxide.
- The process of using a chemical equation to calculate the relative masses of reactants and products involved in a reaction is called stoichiometry
- Thionyl chloride, which is used as a drying agent in many synthetic chemistry experiments, reacts with water to form sulfur dioxide gas and hydrogen chloride gas: SOCl₂(l) + H₂O(l) ►SO₂(g) + 2HCl(g)
- Calculate the mass of water consumed by the complete reaction of 3.50 x 10²g of thionyl chloride.
- Calculate the mass of hydrogen chloride produced by the complete reaction of 3.50 x 10²g of thionyl chloride
Tuesday February 23
AGENDA:
- Hydrofluoric acid, an aqueous solution containing dissolved hydrogen fluoride, is used to etch glass by reacting with the silica, SiO₂, in the glass to produce gaseous silicon tetrafluoride and liquid water. The unbalanced equation is: HF(aq) + SiO₂(s) ►SiF₄(g) + H₂O(l)
- Calculate the mass of hydrogen fluoride needed to react with 5.68g of silica Hint: Think carefully about this problem. What is the balanced equation for the reaction? What is given? What do you need to calculate?
- Calculate the mass of water produced in the reaction described in number 1.
- Methyl Alcohol
- Southern California is famous for may things, and among them, is ______. It is produced when pollutants in the air are trapped near the ground and are caused to react by sunlight.
- A step being considered by the state of California to help solve the smog problem is to replace _____ aka "petrol" with methyl alcohol (usually called methanol).
- Methanol reacts more nearly completely with _____ in a car's engine, thus releasing lower amounts of unburned fuel into the atmosphere. It also produces less _____ (CO(g)) in the exhaust which also encourages the formation of _____ (NO₂(g)), a redish-brown gas that leads to ozone formation and _____ rain.
- Methanol is the only fuel allowed in the open-wheeled race cars used in the _____ 500 and in similar races. It has a higher _____ rating and therefore reduces engine knocking.
- One drawback to methanol is the lower fuel _____. It has high affinity for water which leads to increased corrosion of fuel tank and lines so more expensive stainless steel parts are needed. You need twice the amount of methanol to go the same distance as gasoline however the price to produce it is half of gasoline so its net cost is about the same for both fuels.
- But methanol tends to form _____ (HCHO) when it is combusted and that is a carcinogen and leads to smog as well. Even though California has operated several hundred vehicles on methanol since 1980.
|
mileage
octane gasoline |
nitrous dioxide
smog |
petrol
formaldehyde acid |
carbon monoxide
Indianapolis |
Wednesday February 24
AGENDA:
- For Project points today you will do a report about an animal at the zoo that you saw and relate it to chemistry. I don't want two people doing the same animal. You can do google searches for this on your computers. And I want at least one good page written to be turned in by the end of class. For example, if there were dogs at the zoo, I know that dogs have good smell and it is the chemicals in the air that they are smelling. Their sense of smell is so good that they are used to detect drugs, detect certain sicknesses such as cancer, etc... then I would go on and search for more interesting information to add onto this.
- The remainder of class you will work on finishing up what you can with your chapter reviews which will be due, as usual, on the day of the test. Also remember to read about the Methyl alcohol: Fuel with a future article in your book. You will be tested on that as well.
- Ck-12: Conversion between moles and mass link (this is a good link to get practice on)
- Baking soda, NaHCO₃(s), is often used as an antacid. It neutralizes excess hydrochloric acid secreted by the stomach. The balanced equation for the reaction is: NaHCO₃(s)+ HCl(aq) ► NaCl(aq) + H₂O(l) + CO₂(g) Milk of magnesia, which is an aqueous suspension of magnesium hydroxide, Mg(OH)₂, is also used as an antacid. The balanced equation for its reaction is Mg(OH)₂(s) + 2HCl(aq) ► 2H₂O(l) + MgCl₂(aq). Which antacid can consume the most stomach acid, 1.00g of NaHCO₃, or 1.00g Mg(OH)₂?
- First lets rephrase the question to: How many moles of HCl(aq) will react with 1.00g of each antacid?
- Determine what mass of carbon monoxide and what mass of hydrogen are required to form 6.0 kg of methanol by the following reaction CO₂ + 2H₂(g) ► Ch₃OH(l), 6.0kg (
- Remember that molar masses go with grams and not kilograms therefore you will also have to convert kilograms (kg) to grams (g).
- Determine what mass of carbon monoxide and what mass of hydrogen are required to form 6.0kg of methanol by the following reaction: CO(g) + 2H₂(g) ► CH₃OH(l)
- LITHIUM QUESTIONS
- Lithium seemed to have very few uses for the first 150 years after its discovery in 1817. Within the last 30 years, however, lithium has been found to be a powerful psychoactive drug which treats many manic-depressive aka _____ patients effectively.
- The psychological effects of lithium were found by _____ by someone doing research on manic-depressive syndrome.
- Originally Mr. John Cade thought the manic depressive disease might be due to problems in metabolizing uric acid (also known as its common name, _____. Consequently he administered lithium _____ of uric acid along with lithium carbonate to animals. The result was a dramatic calming effect.
- It wasn't until a year later that another doctor discovered that it was _____, and not the uric acid, that caused the dramatic effects.
- Lithium_____ produces a beautiful _____ flame (looking for a color).
|
ion
urine |
accident
lithium |
salts
|
bipolar
red |
Friday February 26
- AGENDA: Section 9.6: The concept of limiting reactants.
- Using real-life examples with limiting reactants (sandwiches) and concrete analogies (making sandwiches) to help students better understand the calculations. Problems in which reactants are limiting seems to be a great difficulty with students in general.
- 2 pieces of bread + 3 slices of meat +1 slice of cheese ► 1 sandwich
- You have the following ingredients: 20 slices of bread, 24 slices of meat, and 12 slices of cheese
- HOW MANY SANDWICHES CAN YOU MAKE? WHAT WILL BE LEFT OVER? WHAT IS THE LIMITING REACTANT?
- If 249g of methane is mixed with 279g of water, both reactants will "run out" at the same time. The reactants have been mixed in stoichiometric quantities: CH₄(g) + H₂O(g) ► 3H₂(g) + CO(g). On the other hand if 249g methane is mixed with 300g of water, then _____ will be consumed before the _____ runs out. Therefore the _____ will be in excess. Therefore the quantity of products formed will be determined by the quantity of _____ present. Once it is consumed, no more products can be formed, even though there is excess of the other reactant. In this situation, the amount of _____ limits the amount of products that can be formed, and it is the limiting reactant.
- Suppose 25.0 kg (2.50 x 10⁴g) of nitrogen gas and 5.00kg (5.00 x 10³g) of hydrogen gas are mixed and reacted to form ammonia. Calculate the mass of ammonia produced when this reaction is run to completion.
- Aluminum reacts with chlorine gas to form aluminum chloride according to the balanced equation 2Al(s) + 3Cl₂(g) ► 2AlCl₃(s) In a certain experiment, 10.0g of aluminum is reacted with 35.0g of chlorine gas.
- What mass of aluminum chloride will be produced, assuming a complete reaction?
- What mass of which reactant is left after the reaction?
- Nitrogen gas can be prepared by passing gaseous ammonia over solid copper (II) oxide at high temperatures. The other roducgs of the reaction are solid copper and water vapor. How many grams of N₂ are formed when 18.1g of NH₃ is reacted with 90.4g of CuO?
- Iron (III) oxide reacts with CO(g) to form iron metal and carbon dioxide gas: Fe₂O₃(s) + 3CO₂(g) ►2Fe(s) +3CO₂(g) In a certain experiment 5.0g of iron(III) oxide is reacted with 5.0g of carbon monoxide gas.
- What mass of iron will be produced, assuming a complete reaction?
- What mass of carbon dioxide will be produced, assuming a complete reaction?
- Lithium nitride, an ionic compound containing the Li⁺ and N³⁻ ions, is prepared by the reaction of lithium metal and nitrogen gas. Calculate the mass of lithium nitride formed from 56.0g of nitrogen gas and 56.0g of lithium in the unbalanced reaction Li(s) + N₂(g) ► Li₃N(s)
- Using real-life examples with limiting reactants (sandwiches) and concrete analogies (making sandwiches) to help students better understand the calculations. Problems in which reactants are limiting seems to be a great difficulty with students in general.
Monday February 29
AGENDA:
- Finish presentations
- Practice quiz A for chapter 9
- Supernatural powers of animals- 3:07
- E.O. Wilson on Pheromones and the Hidden World of Other Senses- 7:29
- How Ants Comunicate? 2:49
- Male Goats Attract Females With Their Smell- 1:03
- Flowers that look like Animals,Insects and People- 2:51
- Pheromones and Animal Behaviour Communication by Smell and Taste- 1:11
- Alanah and Ella problem #27
- Nick, Gemma, & Taylor #28
Tuesday March 1
AGENDA:
- 1 presentation left
- choose someone from your groups to do problems 27 ad 28
- PLAYED ALL THE PHEROMONE VIDEOS LISTED YESTERDAY SO STUDENTS ARE RESPONSIBLE FOR THIS INFORMATION
- Ella #20, Alanah #21, Nick #22, Taylor #23
Wednesday March 2
AGENDA:
- Dissolving teeth-http://www.ck12.org/chemistry/pH-scale/rwa/Dissolving-Teeth/ real world application of chemistry
- ck-12 Plix chemistry
- Have students come up and do their assigned homework problem
- Go over all sample questions for the last couple days until we are caught up
- Redox overview (ck-12 video)
- ck-12 chemistry practice
- QUIZ (as promised): Thionyl chloride, which is used as a drying agent in many synthetic chemistry experiments, reacts with water to form sulfur dioxide gas and hydrogen chloride gas: SOCl₂(l) + H₂O(l) ►SO₂(g) + 2HCl(g)
- Calculate the mass of water consumed by the complete reaction of 3.50 x 10²g of thionyl chloride. Give answer in significant figures and scientific notation
- Do cola drinks have higher or lower pH values than non-cola ones?
- How much carbon dioxide was found in the soft drink bottle?
- Who first developed a process for artificially carbonating drinks?
- Why is phosphoric acid added to drinks?
- How do soft drinks affect tooth enamel?
- Ella #20, Alanah #21, Nick #22 (do my way), Taylor #23, Gemma & John #24 - show all work in the way that I work it in class and for full credit you should be able to demonstrate it in class
- EXTRA QUESTIONS TO THINK ABOUT FROM MR. ROSEN- Where does most of the acidity of soda pop come from? (for extra credit if you bring it up during class and this question will probably be on the test) Which is more acidic, flat or regular soda, or almost no difference? Which was found to be the most acidic cola with pH of 2.387? Which was found to be the least acidic with pH of 4.038? Which is more erosive, colas or fruit juices? Which is the worst acid for erosion of teeth?
Friday March 4
AGENDA:
- Self-Assessment quiz- Pearson
- Went over ck-12 plix of some old concepts - review
- Handed out STOICHIOMETRY REVIEW HANDOUT ( Fill in what you know and we will go over each and every problem and afterwards take the test- probably end of next week)
Monday March 7
AGENDA:
- GO OVER WORKSHEET
- How can you determine the quantities of reactants and products in a chemical reaction? You use the balanced equation
- Quantity usually means the _____ of a substance expressed in grams or moles? amount
- Stoichiometry is the calculation of quantities in chemical reactions (true/false)? True
- A bookcase is to be built from 3 shelves (Sh), 2 side boards (Sb), 1 top (T), 1 base (B), and 4 legs (L). Write a "balanced equation" for the construction of this bookcase. 3Sh + 2Sb + T + B + 4L = Sh₃Sb₂TBL₄
- From what elements is ammonia produced? How is it used? Ammonia molecules are composed of nitrogen and hydrogen; it is used as a fertilizer
- What kind of information can you NOT get from a chemical equation? moles; mass; size of particles; volume; number of particles
- The coefficients of a balanced chemical equation tell you the relative number of moles of _____ and _____ in a chemical reaction. reactants; products
- Why is the relative number of moles and reactants and products the most important information that a balanced chemical equation provides? Knowing the relative number of moles allows you to calculate the amounts of reactants and products
Tuesday March 8
AGENDA:
- CONTINUE WITH WORKSHEET BELOW
- A balanced chemical equation must obey the law of conservation of mass, true or false?
- The following are always conserved in every chemical reaction?
- volume of gases
- mass
- formula units
- moles
- molecules
- atoms
- What reactant combines with oxygen to form sulfur dioxide? Where can this reactant be found in nature?
- What is essential for all calculations involving amounts of reactants and products?
- The coefficients from a balanced chemical equation are used to write conversion factors called _____.
- What are mole ratios used for?
- True or false, if a sample is measured in grams, molar mass can be used to convert the mass to moles?
- In any problem relating to stoichiometric calculations the given quantity is first converted to _____
- For the equation CH₄(g) + 2O₂(g) ⎆ CO₂(g) + 2H₂O(g) what are the 3 conversion factors you would use to find the volume of carbon dioxide obtained from 1.5L of oxygen?
- What is a limiting reagent?
- True or false, a chemical reaction stops before the limiting reagent is used up?
- The reactant that is not completely used up in a chemical reaction is called the _____
- 2H₂ + O₂ ► 2H₂O Which reactant is the limiting reagent and why?
- What is the theoretical yield?
- The amount of product that actually forms when a chemical reaction is carried out in a laboratory is called the _____ yield.
- Percent yield = _____/______ x 100%
- Describe four factors that may cause percent yields to be less than 100%
- How many moles of oxygen are required to react completely with 14.8 moles of aluminum? 4Al(s) + 3O₂(g) ⎆ 2Al₂O₃(s)
- What is the given information?
- What is the unknown?
- What conversion factor will you need to use
- Complete the solution. 14.8 _____ x 3 mol O₂/_____ = _____ mol O₂
- Why does the answer have three significant figures?
- How many moles of aluminum oxide are formed when 0.78 moles of oxygen rect with an excess of aluminum?
- What information is given
- What information is unknown
- Complete the solution: _____ mol O₂ x ( _____ mol Al₂O₃ / _____) = _____ mol Al₂O₃
- Why does the answer have two significant figures?
- How many molecules of oxygen are produced by the decomposition of 1225 grams of potassium chlorate (KClO₃)?
- The equation for the combustion of carbon monoxide is 2CO(g) + O₂(g) ⎆ 2CO₂(g). How many liters of oxygen are needed to burn 10 liters of carbon monoxide? (Hint: 1 mol of any gas = 22.4 L of the same gas)
- The equation for the complete combustion of ethene (C₂H₄) is: C₂H₄(g) + 3O₂(g) ⎆ 2CO₂(g) + 2H₂O(g)
- If 2.70 moles of ethene reacted with 6.30 moles of oxygen, identify the limiting reagent
- Step 1: Calculate the number of moles of oxygen needed to react with 2.70 moles of ethane. Multiply by the mole ratio
- Step 2: Compare the number of moles of oxygen needed to the number given. (Is the oxygen given equal to or more than amount needed?)
- Identify the limiting reagent
- Calculate the number of moles of water produced.
- Identify the mole ratio needed
- Calculate the given number of moles of oxygen.
- When 84.8 grams of iron (III) oxide reacts with an excess of carbon monoxide 54.3 grams of iron are produced. Fe₂O₃ (s) + 3CO₂ (g) ⎆ 2Fe(s) + 3CO₂(g). What is the percent yield of this reaction?
- Step 1: First calculate the theoretical yield. Begin by finding the molar mass of Fe₂O₃.
- Step 2: Calculate the number of moles of iron (III) oxide. Multiply by the mole/mass (molar mass) conversion factor.
- Step 3: Find the number of moles of Fe expected. Multiply by the mole ratio.
- Step 4: Find the mass of iron that should be produced. Multiply by the molar mass conversion factor.
- Step 5: Compare the actual yield to the theoretical yield by dividing.
- Step 6: Write the answer as a percent, with the correct number of significant figures.
Wednesday March 9
AGENDA:
- SOLUBILITY LAB - 8 SOLUTIONS TO MAKE ONE REACTION WITH A PRECIPITATE PRODUCT AND ONE REACTION WITH ONLY AQUEOUS PRODUCTS
- TEST WILL BE NEXT FRIDAY
Friday March 11
AGENDA: MAKE-UP SOLUBILITY LAB
- You are given 8 different aqueous solutions: Zinc nitrate, sodium sulfate, calcium nitrate, sodium hydroxide, copper nitrate, ammonium hydroxide, potassium iodide, and lead nitrate.
- Your goal is to pick two different solutions and use the solubility rules (page 218 in text) to determine if the product(s) will end up as a solid precipitate or another aqueous solution.
- First without touching the solutions you have to PREDICT a combination that will make a solid precipitate product and then you have to PREDICT another two solutions when combined with each other will not precipitate anything (all aqueous products).
- Once you have made your predictions then you will come up and show me the two equations and if they are made correctly (balanced equations, correct oxidation numbers, subscript numbers, naming of metal with correct polyanion, cations of one reactant combining with anion of the other, etc... ) then you will actually mix the two solutions together (a drop of each into a depression of a reaction tray)
Monday March 14
AGENDA:
- Finish up the lab
- Go over review
- VINEGAR = DILUTED ACETIC ACID
- BAKING SODA =
- KOSHER SALT =
- CALCITE =
- BLEACH =
Tuesday March 15
AGENDA:
HOMEWORK:
HOMEWORK:
- Chapter 9 Review Questions #35: Alanah #b, Nick #c, Ella #d
Wednesday March 16
AGENDA:
- LABORATORY: pH Measurements/Indicator with methyl orange, brom thymol blue and phenolphthalein
- Methyl orange (liquid): Usually used in titrations for acids. It is an orange color which turn redder in acidic solutions and yellower in basic solutions.
- Bromothymol blue:
- Phenolphthalein
- Universal Indicator
- The indicator reaction is pH dependent because it involves either the release or capture of hydrogen ions. (HIn) ⑅ (H⁺ + In⁻) where "HIn" is the indicator attached to hydrogen ion and "H⁺ + In⁻" is without an attached hydrogen ion. The two forms of the indicator molecule have noticeably different colors. For example, bromothymol blue has a yellow HIn form and a blue In form. When there are equal amounts of HIn and In, the solution looks green. Adding a drop of acid adds H+ions which react with the In- ions to form HIn, and the solution becomes more yellow. Adding a drop of base converts HIn to "In⁻ + In", and the solution becomes more blue.
But what exactly does attaching a hydrogen to the molecule do to cause a color change? To understand the answer, you will have to know a little about how color, molecular structure, quantum levels of the electron, photons, and the physics of color absorption and reflection relate to one another. Explained here.
Friday March 18
AGENDA:
- TEST TODAY:
- YOU ARE EXPECTED TO INCLUDE ALL UNITS (EVEN IN YOUR CALCULATIONS), ALL STATES OF MATTER [(s), (g), (aq), (l)], SIGNIFICANT FIGURES AND YOUR ANSWER SHOULD BE WRITTEN IN SCIENTIFIC NOTATION UNLESS OTHERWISE STATED.
- Ag(s) + H₂S(g) ⎆ Ag₂S(s) + H₂(g) Balance this equation and then identify the mole ratio that you would use to calculate the number of moles of Ag(s) needed to react exactly with a given number of moles of H₂S(g).
- What quantity serves as the conversion factor between the mass of a sample and the number of moles this sample contains?
- Calculate the number of moles of iron (II) sulfate in 12.7 mg of iron (II) sulfate.
- 2 Mg(s) + O₂(g) ⎆ 2 MgO(s) is the balanced equation for photographic flash reactions using magnesium metal. How many grams of MgO(s) are produced by the complete reaction of 1.25 g of magnesium metal?
- K(s) + I₂(s) ⎆ KI(s) For this unbalanced equation suppose that exactly 15.0g of each reactant is taken. Determine which reactant is limiting, and calculate what mass of product is expected. (Assume that the limiting reactant is completely consumed)
- EXTRA CREDIT: The article in your textbook talks about trying to solve the smog problem in California by replacing gasoline with what?
- EXTRA CREDIT: What was the featured celebrity chemical in this chapter?
- EXTRA CREDIT: The "Golden" Dollar article in this chapter depicts this famous Shoshone Indian woman on the front?
Tuesday March 29
OBJECTIVES:
- Understand the general properties of energy
- Understand the concepts of temperature and heat
- Consider the direction of energy flow as heat
- Understand how energy flow affects internal energy
- Understand how heat is measured
- Consider the heat (enthalpy) of chemical reactions
- Understand Hess's Law
- See how the quality of energy changes as it is used
- Consider the energy resources of our world
- Understand energy as a driving force for natural processes
- Read 10.1 to 10.4 and answer section 10.1 - 10.4 questions
- Work is done when a force is done to move an objecT
- Answer chapter review questions 1-14
- Potential energy is due to position or _____. Kinetic energy is due to an object's _____.
- Work is is force acting over a _____
- All energy is not available to produce work because some of it is given off as _____ or _____
- A _____ is something that is not dependent on the pathway. (heat/energy/work) _____ is an example of one. Another example is _____.
- _____ is a flow of energy due to a temperature difference.
- Temperature is a measure of average _____
- Thermodynamics is the study of _____ and its changes.
- In an _____ process, energy as heat flows from the surroundings into the system. In an _____ process, heat flows out of the system into its surroundings.
- State function, out of energy, heat and work, which are state function(s)?
- Potential energy in a chemical reaction is stored where?
- What is the study of energy called?
- The _____, E, of a system can be defined most precisely as the sum of the kinetic and potential energies of all "particles" in the system. It can be changed by a flow of _____, heat, or both
- △E = q + w is an equation where △ ("_____") means a change in the function that follows and q stands for _____ and w stands for _____.
- Thermodynamic quantities always consist of two parts: a number and a sign, indicating the direction of the flow. The sign represents the _____ 's point of view and not the _____'s point of view.
- If a quantity of energy flows into the system via heat (an _____ process)), q would be positive which indicates that the system's energy is increasing. The same conventions apply to the flow of work.
- If a chemical has a low potential energy then the chemical is stable and the _____ holding the atom in the molecule are strong.
- In an endothermic reaction the reactants have the (lower/higher) potential energy?
- When energy is reported with a negative sign (△E < 0) then that means that it is an (endothermic/exothermic) reaction.
- When work is reported with a positive sign then the (system/surrounding) does work on its (system/surroundings)
- Heat is one way that _____ is transferred from one object to another.
- Thermal energy is the energy that a substance contains due to the random _____ of its components.
- When we heat a substance to a higher _____, we increase the motions of the components of the substance, its thermal energy.
|
chemical bonds
delta thermal energy endothermic thermodynamics motion |
internal energy
kinetic energy distance motions surrounding light |
system
energy work heat composition state function |
displacement
bonds exothermic heat pathway |
Wednesday March 30
AGENDA: ENERGY AKA THERMODYNAMICS
- Go over 10.1 to 10.3
- Explanation of last lab (acids and bases)
- LAB: Identifying the unknown solution: You have 3 unknowns in front of you (HCl, NaOH and CuSO₄ which is on the acidic side). You also have a table of solubility and Red and Blue litmus paper. You will be able to identify one of them and tell me what kind of cation you would need to distinguish the other two.
- Your answers will make a difference on the grading of your lab because you already should have the knowledge needed to know the answers
- All chapter review problems correlating to 10.4 (that would be 15- 21)
Friday April 1
April Fool's Day
AGENDA:
- Went over fill-ins and homework
- Went through all of the textbook examples of 10.5 therefore homework should just be review.
- Start working on chapter review problems for 10.5 (22-28)
- Review problems for section 10.5 (22-28) all
- Start reading section 10.6
- The voodoo lily is a beautiful and seductive plant but it smells terrible. Despite its antisocial odor, this putrid plant has fascinated biologists for many years because of its ability to generate _____. The plant's blossom can be as much as 15 ℃ above its surrounding temperature.
- The lily prepares a malodorous mixture of chemicals characteristic of rotting meat which attract flesh-feeding beetles and flies. Once the insects enter the _____chamber, the high temperature there cause the insects to remain very active to better carry out their duties.
- The voodoo lily smells so bad it's nickname is "The _____ Flower". It also produces the largest flower in the world which is called _____, the largest having reached the height of 6 meters (or _____ feet).
- So the connection between biology and chemistry here is that this plant (biology) produces a smell (chemistry) which attracts the kind of insects it needs to aid with pollination and leave offspring.
- FIREWALKING: MAGIC OR SCIENCE?
- Human tissue is largely composed of_____, which has a relatively large _____. This means that a large amount of _____ must be transferred from the coals to change significantly the temperature of the feet. During the brief contact between feet and coals involved in firewalking, there is relatively little time for energy flow, so the feet do not reach a high enough temperature to cause damage. Also, although the surface of the coals has a very high temperature, the red-hot layer is very thin. Therefore, the quantity of evergy available to heat the feet is smaller than might be expected .Thus, although firewalking is impressive, there are several scientific reasons why anyone with the proper training should be able to do it on the properly prepared bed of coals
- CELEBRITY CHEMICAL: METHANE
- Methane is the main component of _____, a valuable fossil fuel. An abundant supply of it lies in the depths of the ocean.
|
pollination
water |
corpse
natural gas |
Twenty feet
specific heat capacity |
heat
|
Titan Arum
energy |
Monday April 4
AGENDA:
- Go over homework and then i will do sample exercises in the book
QUESTIONS: SECTIONS 10.5 -
- In the metric system the _____ is defined as the amount of energy (heat) required to raise the temperature of one gram of water by one Celsius degree
- The "calorie" which you are more familiar with is used to measure the energy content of food and is actually kilocalorie, written with a capital C (Calorie). The _____ (an SI unit for energy) can be defined by (1 cal = 4.184 J)
- The _____ of a substance (the energy required to change the temperature of one gram of the substance by one Celsius degree) is used to calculate temperature changes when a substance is heated.
- The change in _____ for a process is equal to the heat for that process run at constant pressure.
- This allows for the calculation of the heat of a given reaction from known heats of related reactions.
- I want to know how many calories are in 20 Calories? Answers a) trick question b) 20 c) 20,000 d) They can't be converted with each other.
- How does water relate to other substances in relation to specific heat capacity? a) it is just the set point b) high c) low d) average
- Knowing that the specific heat capacity of water is really high compared to other substances what does this mean? It means that if you happen to live by an ocean or other large body of water than the temperature around you will be a lot more _____ than most other places.
- Water has a unique ability to absorb _____. Therefore it has a moderating influence on the coastal regions and keeps the temperature within a (high/low) range of temperature. One of water's most useful properties is its ability to act as a _____. Also, nuclear power plants use vast quantities of water to carry away the excess heat of the nuclear fission process.
- The following equation: Q = s x m x △T where Q= Energy (heat), s = specific heat capacity, and △T = _____ in temperature in degrees _____
- The symbol △ stands for the Greek letter _____
- Exothermic reactions _____ heat energy while endothermic reactions _____ heat energy.
- Scientists have invented a special energy function called _____ which is designated by H. For a reaction occurring under conditions of constant _____, the enthalpy change for a reaction is the same as the _____ for that reaction.
- How are milliliters, cubic centimeters (cc) or (cm³), and grams related to one another? What does a milliliter measure? What does a cubic centimeter measure? What does a gram measure?
- △H⍴ = _____ where the subscript p indicates that the process has occurred under conditions of constant pressure.
- A _____ is a device used to determine the heat associated with a chemical reaction
|
enthalpy
calorie delta Hess's law mass |
pressure
produce joule moderate |
coolant
specific heat capacity heat absorb |
volume
enthalpy change Celcius |
Monday April 4
AGENDA continued:
- Calorimetry: Crash course chemistry #19 - 11:56
- 10.6 The equation for the fermentation of glucose to alcohol and carbon dioxide is C₆H₁₂O₆ (aq) ⎆ 2CH₅OH(aq) + 2CO₂(g) The enthalpy change for the reaction is -67 kJ. Is the reaction endothermic or exothermic? Is energy, in the form of heat, absorbed or released as the reaction occurs? Here the problem looks harder than it actually is. It is conceptual. If you see that the enthalpy change (heat released or absorbed) is negative then you know that heat is released and it is an exothermic reaction
- HOMEWORK: chapter review problems 30 to 33; Read 10.8
Tuesday April 5
AGENDA: TEST FRIDAY
QUESTIONS;
- Go over fill-ins during class
- Focus question #4 on page 305: The enthalpy of combustion of solid carbon to form carbon dioxide is -393.7 kJ/mol ℃, and the enthalpy of combustion of carbon monoxide to form carbon dioxide is -283.3 kJ/mol ℃. Using these data, calvulate the change in enthalpy for the reaction 2C(s) + O₂(g) ⎆ 2CO(g)
QUESTIONS;
- True or false: Enthalpy is a state function.
- In going from a particular set of reactants to a particular set of products, the change in enthalpy is the same whether the reaction takes place in one step or in a series of steps. This principle is known as _____. The importance of this is that it allows us to calculate heats of reaction that might be difficult or inconvenient to measure directly (such as in a calorimeter).
- The _____ can measure energy changes.
- Enthalpy is the heat absorbed or given off for a reaction. It must have a magnitude as well as a _____.
- A negative heat of reaction means that energy (leaves/enters) the system)
- To find the heat of reaction the mass must be multiplied by the specific heat capacity and the result multiplied by the change in _____.
- One of the most important characteristics of _____ is that it is conserved.
- The energy stored in the _____ of gasoline (C₈H₁₈ and other similar compounds) and of the oxygen that reacts with it is changed to _____ energy during the driving of a car.
- When we utilize energy to do work, we degrade its usefulness. In other words, when we use energy the _____ of that energy (its ease of use) is lowered. In summary concentrated energy (eg gasoline) is used easily when combusted with oxygen to drive a car but now all that energy is heat which is spread throughout the drive which can't really be used.
- The energy stored in _____ molecules got there over millions of years through plants and simple animals absorbing energy from the sun and using this energy to construct molecules. It is highly valuable because it furnishes a convenient, concentrated source of energy. Unfortunately, we are using this fuel at a much faster rate than natural processes can replace it. It is a thick, dark liquid composed mostly of compounds called _____ that contain carbon and hydrogen
- By the process of photosynthesis, plants store energy that can be claimed by burning the plants themselves themselves or the decay products that have been converted over millions of years to _____
- Petroleum and _____ , consisting mostly of methane but also ethane, propane, and butane, were formed from the remains of marine organisms that lived a long time ago.
- In the mid 1800s _____ was the main source used for energy.
- Petroleum in the 1800s was refined to produce _____ which served as an excellent lamp oil. Gasoline had limited use and was often discarded.
- As gasoline became more important, new ways were sought to increase the yield of gasoline from petroleum. A process called pyrolytic _____ is a process where the heavier molecules of the kerosene fraction are heated to about 700 ℃, causing them to break (crack) into the smaller molecules of hydrocarbons in the gasoline fraction. As cars became larger the engines started to "knock" and produce unwanted noise and engine damage. Intensive research was done to find additives that would promote smoother burning and an "antiknock" agent was found called tetraethyl _____. This was bad for the environment and that is why we have _____ gasoline now.
- _____ is another fossil fuel. It was formed from the remains of plants that were buried and subjected to high pressure and heat over long periods of time. It is more plentiful than petrol products.
- The atmosphere, like window glass, is transparent to visible light but does not allow all the _____ radiation to pass back into space. Molecules in the atmosphere, principally _____ and _____, strongly absorb infrared radiation and radiate it back towards the earth. A net amount of thermal energy is retained by the earth's atmosphere, causing the earth to be much warmer than it would be without its atmosphere. In effect, the atmosphere traps some of the energy, acting like the glass in a greenhouse keeping the earth warmer than it would otherwise be. This is called the _____ effect.
- The increase in the atmospheric concentration of _____ is quite dramatic.
- One possible alternative to petroleum as a source of fuels and lubricants is _____
- Are petroleum products really nonrenewable? Forget what you've heard!
- These are the two most important driving forces that causes events to occur
- _____, symbolized by "S", is the measure of disorder or randomness
|
energy spread
carbon dioxide unleaded natural gas petroleum temperature |
greenhouse
coal hydrocarbons deHess's Law entropy thermal |
vegetable oil
infrared kerosene calorimeter energy |
matter spread
water lead sign fossil fuels |
Wednesday April 6
AGENDA:
- Quiz A
- Review Handout - go over and give answers
- fill-ins review
Friday April 8
AGENDA: CHAPTER TEN TEST
- Express 88.2 Joules in calories
- A sample of water requires 180J to be heated 5.7 ℃. What mass of water is present?
- When 1 mole of methane (CH₄(g)) is burned at constant pressure 890 kJ are released as heat. What amount of heat would be released if a 100.0g sample of methane is burned?
- Even though there is an "energy crisis," the total quantity of energy conserved. Is this true or false? If true then what can you add?
- The function that is related to the disorder-matter-spread is called what? What is its symbol?
- What kind of reactions absorb heat from the surroundings? Would the magnitude of this heat be negative or positive?
- Name all the state functions that you can.
- What is the device called that can measure energy changes?
- After filling up your car with 8 gallons of gasoline and driving around for 88 minutes what has happened to the gasoline that is no longer in the gas tank?
- EXTRA CREDIT: Where is potential energy stored in chemical reactions?
- EXTRA CREDIT: What are compounds called that contain just carbon and hydrogen?
- EXTRA CREDIT: What was the main source used for energy in the mid 1800s?
- Do the reactants or the products have the lower potential energy in an endothermic reaction?
- Boiling water is an (endo/exo-thermic) reaction? Because why?
- Express 108 calories in Kilojoules
- A gas absorbs 45 kJ of heat and does 29 kJ of work. Calculate the change in energy.
- How much energy (J) is required to heat 2.50 g of water from 21.0℃ to 26.5℃?
- A sample of water requires 180 J to be heated 5.7 ℃. What mass of water is present?
- When we use energy what are the byproducts? What is lowered?
- EXTRA CREDIT: What are compounds called that contain just carbon and hydrogen?
- EXTRA CREDIT: In the symbol (Hp, the p stands for) (a) Partial enthalpy (B) Constant pressure (C) Variable pressure (D) heat of reaction
Monday April 11
OBJECTIVES: (a lot of review)
- Describe atomic orbitals (s,p,d,f) and their basic shapes
- Describe characteristics electromagnetic radiation: waves, wavelength, frequency, energy and speed.
- Describe and explain the organization of elements into periods and groups in the periodic table
- Use the periodic table to determine the atomic number, atomic mass; mass number; and number of protons, electrons, and neutrons in isotopes of elements
- Calculate the weighted average atomic mass of an element from isotopic abundance, given the atomic mass of each contributor
- Identify regions (e.g. groups, families, series) of the periodic table and describe the chemical characteristics of each
- Use the periodic table to predict and explain the valence electron configurations of the elements.
- Understand how atoms emit light
- Understand electron spin
- Understand how principal energy levels fill with electrons in atoms beyond hydrogen
- STARTING CHAPTER 11: "MODERN ATOMIC THEORY"
- Assignment for today. You will get your tests back and you will work out the problems that you got wrong and hand them in all corrected by the end of class.
- I will start making dates for make up tests because I am noticing that the students in this class will postpone it and continue to postpone it until the last minute, probably right up until finals, and that isn't a good way to learn chemistry. Therefore I'm going to have to set deadlines to push you into studying. Therefore I'm going to make the make-up tests available only on Wednesday block days. However this Wednesday I will be gone for a field trip.
- The differences in behavior between groups of atoms in the periodic table is because of the way _____ are arranged.
- Energy is transferred from one place to another by light. This is more properly called _____
- The _____ is the distance between two consecutive wave peaks. The _____ of the wave indicates how many wave peaks pass a certain point per given time period.
- Whenever atoms receive energy from some source they become excited. They can then release this energy by releasing _____.
- The emitted energy is carried away by a _____. High-energy photons correspond to _____-wavelength light and low-energy photons correspond to _____-wavelength light.
- When a plant senses red light then the plant knows it has many neighbors because the plants around it are _____ red light (the plants are green). Because these neighbors are competitors for light the plant directs its growth above ground, producing a taller, thinner structure that can compete more successfully for sunlight. For opposite effect, orange is used to cover turnips in soil as it reflects red light which signalled "no competition" and encouraged root growth rather than shoot growth resulting in bigger turnips (root).
- The _____ of a wave is how fast the wave travels through a medium while the _____ of a wave is the number of waves passing a particular point in a given time.
- The relationship between energy and wavelength is _____. The longer the wavelength, the less energy (and less frequency) is in its photons.
- Plants see _____ light as meaning that many other plants are competing for sunlight. This causes the plant to grow above ground quicker, resulting in faster seedling growth and earlier, larger _____.
|
electrons
short fruit long |
reflecting
speed frequency |
red
inverse electromagnetic radiation |
wavelength
photons light |
|
|
Wednesday April 13
AGENDA:
- Students should have already read sections 11.1 to 11.3 so answer focus questions on page 327
- Also during class read sections 11.4 to 11.6 (they are only 1 page each) and answer focus questions on page 332
- Then you will be responsible for Chapter review problems 1 to 23 due Friday
- There will be a quiz on 11.1 to 11.6 on Friday
- "IF" done with all of the above then see if you can answer the fill-in questions I asked on this Monday (this will help with 11.1 to 11.3)
- "IF" done with everything then scroll up to March 18 and re-do your test and I want it turned in with all the right answers on it.
- Animations for chapter 11 videos for chapter 11
- Chapter quizzes
- Quiz A
- Quiz B
- Quiz C
Monday April 15
AGENDA:
- Go over electromagnetic radiation again
- An atom of excess energy is said to be in an _____ state. It can release some or all of its excess energy by emitting a photon and move to a lower energy state. The lowest possible energy state of an atom is called its _____ state.
- The energy levels in all atoms have only certain discrete values of energy and not a continuous range. This means that the energy levels in atoms are _____. A useful analogy for this is the contrast between the elevations allowed by a ramp and those allowed by a set of steps. The quantized nature of atoms is a surprise and also nonintuitive.
- _____ lights produce a continuous spectrum because the tiny filament is radiating all wavelengths of visible light whereas vapor lamps are similar to gas discharge tubes.
- _____ vapor lights have a blue cast, _____ lights are yellow, _____ gives a a deep-red color, _____ burns a green color, and _____ lights are more orange.
- The atom pictured as a small positive nucleus with electrons orbiting around it like the planets orbiting the sun is the _____ model of the atom. This is fundamentally incorrect.
- There is now much evidence suggesting that all matter exhibits both _____ and _____ properties.
- An _____ describes the areas around the nucleus where an electron is likely to be found.
- _____ is the most abundant element in the universe. Liquid hydrogen is used to power the _____
- Hydrogen seems like an ideal fuel because it provides lots of _____ per gram and it gives only _____ as a combustion product instead of the _____ produced by fossil fuels. However, there are two major problems with using hydrogen as a fuel. First, virtually no _____ hydrogen exists, so energy must be expended to free it from compounds such as water. Second, hydrogen has a very low boiling point and requires very low temperatures and very high pressures to store as a liquid.
- Each atom has _____ energy levels that are labelled with integers. Each of these is subdivided into sublevels represented by a letter. The _____ tells the principal energy level and the letter tells the _____.
- The first sublevel is represented by the letter ____ and the second represented by the letter ____, principal level 3 has three sublevels, the third of which is represented by the letter ____, and principal level 4 has four sublevels the 4th of which is represented by the letter ____.
- Principal level 1 consists of one sublevel which contains the _____ orbital. principal level 2 has two sublevels which are the _____ orbital and the three _____ orbitals. Principal level 3 has all of level two plus five _____ orbitals. Principal level 4 has all of level three plus 7 _____ orbitals.
- The electron has a special property that determines how they can be arranged in an atom's orbitals. This property is _____. Two electrons must have _____ spins to occupy the same orbital. If they have the same spin they cannot be in the same orbital. An atomic orbital can hold a maximum of _____ electrons and they must have opposite spins. This is called the _____
- As "n" increases (n= principal energy level), the shape of a given orbital stays the same while the _____ increases.
- Three dimensional probability maps that represent the likelihood that the electron will occupy a given point in space are known as _____.
- A set of orbitals of a given type of orbital within a principal energy level is called a _____.
- An _____ is a particular path for something revolving around something else. An _____ describes a region of probability for finding an electron.
|
orbit
spin free f excited wave space shuttle |
mercury
principal 1s Pauli exclusion principle energy quantized Bohr |
shape
water 2p neon orbitals incandescent particle |
2s
orbital sodium ground d sublevel carbon dioxide |
Monday April 18
AGENDA:
- Reviewed everything up including 11.8
- SECTION 11.9 For the first 18 elements, the individual sublevels fill in the following order: 1s, then 2s, then 2p, then 3s, then 3p
- MAKE UP TEST FOR CHAPTER 10 AFTER BREAK
- CHAPTER REVIEW QUESTIONS 11.7 & 11.8 (24 to 33)
- READ SECTION 11.9 (You should be able to give the electronic configurations and orbital diagrams of the first 18 elements on the periodic table of elements)
- Dmitri Ivanovich Mendeleev (1834-1907) was born in Siberia, the youngest of SEVENTEEN (17) children, taught _____ at the University of St. Petersburg, wrote a book on _____ chemistry in 1861 and then later a book on _____ chemistry. He was struck by the fact that the systematic approach for characterizing organic chemistry was lacking in inorganic chemistry. In attempting to systemize inorganic chemistry, he ended up creating the _____.
- If you were placed inside a strong magnetic field (such as inside the hollow core of an electromagnet), your cells would respond by moving in a fashion that produces magnetic fields aligned to oppose the inducing field. This phenomenon is called _____.
- The University of Nijmegan, the Netherlands, have levitated frogs, grasshoppers, plants, and water droplets, among other objects. Given a large enough _____ even humans can be levitated. But that would be very expensive.
- Most of the signals in an _____ scan originate from the hydrogen atoms in water. The difference in the way water is bound in different _____ produces the signal variations between healthy and diseased tissue. Diseased tissue produces a distinctive MRI image. MRI can also produce clear images of _____
- The electrons that are of the most importance to chemists are located in the outermost (highest) principal energy level of an atom and are called _____ electrons. They are the ones that attach to each other to form bonds and elements with the same valence electron arrangement show (in the same group) very similar chemical behavior. The inner electrons, or _____ electrons, are not involved in bonding atoms to one another .
- The elements that correspond to filling the 3d orbitals are called _____
|
blood vessels
chemistry diamagnetism |
Periodic table of the elements
MRI valence |
core
organic chemistry electromagnet |
tissues
inorganic chemistry |
Wednesday April 20
"Drug Dog Day 4/20 inspection!!"
AGENDA:
- TEST KAHOOT TOMORROW (also retake chapter 9)
- Going over electronic orbital configurations
- Handed out electronic orbital worksheet (if turned in next class period complete you will get extra credit)- if you were absent you can download it below "Electronic Orbital Configurations"
- Please use the following (upper and lower charts) to your advantage
| electronic_orbital_configurations.pages | |
| File Size: | 906 kb |
| File Type: | pages |
Monday April 25
AGENDA:
- Went over electron configuration (long version and abbreviated version) due tomorrow
Tuesday April 26
AGENDA:
QUESTIONS:
- The elements Cu and Cr are exceptions to the expected electron configuration. The reasons for these exceptions are quite complicated. Cr instead of being 3d⁴ would be 4s¹3d⁵ and Cu instead of being 3d⁹ would be 4s²3d¹⁰
- With an understanding of the structure of the periodic table, you can figure out the expected electron configuration of any element. You don't need to memorize these configurations
QUESTIONS:
- The shape of an orbital does not indicate the details of electron _____. It indicates the _____ distribution for an electron residing in that orbital.
- The electron is somewhere inside the orbital surface _____ of the time.
- Atoms with more than one electron are called _____ atoms.
- Electron configurations can be represented by using an orbital _____ also called a box diagram in which orbitals are represented by boxes grouped by sublevel with small arrows indicating the electrons.
- The abbreviated way of writing electron configurations is called the _____ notation. This places emphasis on the outer electrons used in bonding. In this notation the last noble gas is used and the outer electrons are written after it.
- The elements ______ and _____ are notable exceptions to the expected electron configuration.
- The group label at the top of the periodic table gives the total number of _____ electrons for that group.
- Chemical properties are similar in elements in the same _____ of the periodic table.
- _____ typically have the following physical properties: a lustrous appearance, the ability to change shape without breaking (pulled into a wire or pounded into thin sheet), and excellent conductivity of heat and electricity. _____ typically do not have the above properties (exceptions are that solid iodine is lustrous and graphite form of _____ is an excellent conductor of electricity and diamond form is an excellent conductor of heat)
- The chemical differences between metals and nonmetals are that metals tend to lose electrons to form _____ (positive ions) and nonmetals tend to gain electrons to form _____ (negative ions). When a metal and nonmetal react, a transfer of one or more electrons from the metal to the nonmetal often occurs.
- As we go (up/down) a group, the metals become more likely to lose an electron.
- Atomic radii (increase/decrease) in going from left to right across a period. This can be explained by the increase in _____ (protons) charge. This pulls the electrons closer to the nucleus.
- Atomic radii (increases/decreases) down a group.
- The energy required to remove an electron from an individual atom in the gas phase is called the _____ of the atom. There there is an inverse relationship between the ionization energy of an atom and its atomic radii.
- Ionization energies tend to (increase/decrease) in going from the top to the bottom of a group.
- In contrast to metals, nonmetals have relatively (large/small) ionization energies.
- The most chemically active metals are in the (upper/lower-right/left) region of the periodic table while the most chemically active nonmetals are in the (upper/lower-right/left) region.
- Metals have relatively few _____ electrons located in the outer shell. In contrast, nonmetals have a large number of electrons in the outer shell.
|
valence
period movement anion diagrams |
cation
metals probability core |
nuclear
nonmetals 90% chromium |
ionization energy
carbon polyelectronic copper |
Wednesday April 27
AGENDA:
- TEST TODAY (BOTH CHAPTER NINE RETAKE & CHAPTER 11)
- BOZEMAN SCIENCE- Electron configuration and ionization energy_
- Quiz A
- Quiz B
- Quiz C
- CHAPTER 11 ANIMATIONS
Monday May 2
AGENDA: (Substitute, out sick)
- Start reading as much of chapter 12 as you can (at least 12.1 to 12.3)
- FOCUS QUESTIONS SECTIONS 12.1 - 12.3
- Know all the following questions
- A force that holds groups of two or more atoms together and makes them function as a unit is called a _____. Its strength is found by measuring the energy required to break it which is called it's _____.
- The strongest bonding force present from attractions among oppositely charged ions is called a(n) _____. The type of bonding such as in water where electrons are shared by nuclei is called a(n) _____. Between these two are intermediate cases in which there is an unequal sharing of electrons forming what is called a(n) _____.
- The relative ability of an atom in a molecule to attract shared electrons to itself is its ______. The more of the difference of this property between the bonding atoms the more electronegative it is. _____ bonds have the largest electronegativity.
- If something is polar it has a _____ moment, making it behave as though it contained a negative and positive charge.
- Because water is _____, it can surround and attract both positive and negative ions. This causes ionic materials to _____ in water. It is the polarity of water that makes the requiring of much energy to change from liquid to gas. If water were nonpolar, water would be a gas and oceans would be empty.
- in a _____ covalent bond, the electrons spend more time closer to one atom than to the other. In a _____ covalent bond, the electrons are shared equally between the atoms.
- The _____ elements are those elements within the first two families (Groups I and II on the far left) and the last six families or groups (on the right) of the Periodic Table
- Atoms have a strong tendency to adopt a _____ configuration.
- A new material that is likely to be a boon to auto designers is _____ metal foam. It is lightweight and stiff and also increases the ccrash worthiness of a car due to its energy absorbing abilities.
- A _____ is always smaller than the parent atom, and an _____ is always larger than the parent atom.
- When a molecule such as ammonium nitrate is dissolved in water, it behaves as a strong _____ like the binary ionic compounds. This is because the ions are freed to move independently and can conduct an electric current.
- The _____ of a car is directly related to its fuel economy. The lighter the car, the better the fuel efficiency.
- The Lewis structure is a representation of a molecule that shows how the _____ electrons are arranged among the atoms in the molecule by following the _____ rule. The valence electrons are represented by _____
- The _____ number gives the total number of valence electrons.
- In a Lewis structure a _____ (instead of a pair of dots) is used to indicate each pair of bonding electrons.
- A molecule shows _____ when more than one Lewis structure can be drawn for the molecule.
- The compound that gives pepper its characteristic "hot" taste is called _____. It is used as a deterrent for animals such as for coating boats so barnacles keep from sticking to it, so rodents keep from gnawing on fiber-optics and to prevent pets from disturbing a healing wound.
- Exceptions to the octet rule are _____ and ____ where they both have fewer than eight electrons around it. Another is diatomic _____. It does not agree with its observed behavior as it "sticks" between the poles of a strong magnet making its diatomic form paramagnetic which the regular Lewis is not congruent with.
|
electrolyte
noble gas ionic bond capsaicin octet covalent bond polar covalent bond |
boron
group bond ionic dots cation electronegativity |
weight
aluminum dipole beryllium polar line dissolve |
oxygen
valence anion representative bond energy resonance nonpolar |
Tuesday May 3
AGENDA:
- Continue reading chapter 12.4 - 12.5 and do the focus questions
- If done with this then do the homework
- Read sections 12.6-12.7 and do the focus questions
- Odds or even 1-38 of review questions (only need to answer one of the a,b,c, or d)
Wednesday May 3
AGENDA:HOMEWORK:
- Read over 12.8 - 12.10
- Molecular or _____ structure are terms that refer to the three-dimensional arrangement of the _____ within a molecule
- One can learn to predict molecular geometry from the number of _____
- VSEPR is the valence shell electron pair repulsion model. It is useful for predicting the molecular structures of molecules formed from _____.
- The structure around a given atom is determined by minimizing _____ between electron pairs. This means that bonding and non-bonding (lone pairs) electron pairs around a given atom are positioned as far apart as possible.
- Whenever two pairs of electrons are present around an atom, they should always be placed at an angle of _____ degrees to each other to give a _____ arrangement.
- Whenever three pairs of electrons are present around an atom (0 lone pairs), they should always be placed at the corners of a triangle in a plane at an angle of _____ degrees to each other in a _____ structure.
- Whenever three pairs of electrons are present with one lone pair it has _____ structure
- Whenever there are four pairs of electrons in with all pairs bonding (0 lone pairs) that would put the electron pairs even farther apart at _____ degrees between each other in a _____ arrangement. An example is _____
- Whenever there are four pairs of electrons with 3 pairs bonding (1 lone pair) you have a _____ arrangement. There are _____ degrees between atoms.
- Whenever there are four pairs of electrons with 2 pairs bonding (2 lone pairs) you have ______ structure with _____ degrees separating each atom.
- When using VSEPR to predict molecular geometry, a double bond is counted the same as a single _____. The same can be said about triple bonds.
|
electron pair(s)
180 linear |
bent or angular
methane geometric 120 |
trigonal pyramid
trigonal planar atoms |
109.5
tetrahedral nonmetals repulsions |
| lewis_structure_worksheet_1.pdf | |
| File Size: | 7 kb |
| File Type: | |
Friday May 6
AGENDA:
- Went over how to find out how many valence electrons there are in each element. Take the most outer energy level and add up the electrons in only the outer level "s" and "p" orbitals.
- Went over drawing Lewis structures: 1) count number of total valence electrons. 2) Draw a line which represents two electrons to an atom bound to another atom (sharing a pair of electrons) and subtracting the number of these electrons from the total valence electrons. 3) With the remaining electrons left distribute them accordingly into the the remaining orbital spaces with hydrogen having a maximum of two electrons in its valence shell and all the other elements having 8 electrons maximum in its valence shell
- TO CONVERT PRESSURE 1.000 atm = 760.0 mm Hg = 760.0 torr = 14.69 psi = 101,325 Pa
- King of Prussia in 1683 awarded a lifetime pension to an Otto von Guericke, physicist, who demonstrated for the king two hemispheres together with the air pumped out of them, that teams of horses could not pull the hemispheres apart. Then , after secretly opening the air valve, Guericke easily separated the hemispheres by hand.
- When a balloon is placed in a freezer, its volume is _____.
- A gas uniformly fills any container, is easily _____, and mixes completely with any other gas.
- The meteorologist who says that a "low" is approaching means that the atmospheric pressure is going to decrease and this often occurs in conjunction with a _____.
- As a gas, _____ occupies 1300 times as much space as it does as a liquid at 25 ℃ and atmospheric pressure.
- The most common units of measure are based on the height of the liquid in a column - in mm- that the gas pressure can support. The liquid that we use is _____ because of its high density. At atmospheric pressure the reading is 760 mm Hg and mm Hg is often called the _____ in honor of Torricelli so the terms are interchangeable.
- There are other units used for pressure. Standard atmosphere (abbreviated ____) and 1 of these = 760 mm Hg. But the SI unit for pressure is the _____ abbreviated Pa and 1 standard atmosphere = 101,325 Pa. We also use psi which stands for pounds per square inch.
- Mercury is used in thermometers instead of _____ because instead of going up to 760 mm the liquid will go up to about 34 feet! This is because mercury is about 13.5 times denser.
|
decreased
atm |
storm
mercury |
compressed
pascal |
water
|
Monday May 9
AGENDA:
- Time to catch up and do chapter review questions up to 12.7
- When done do the VSEPR handout http://misterguch.brinkster.net/PRA024.pdf
- VESPR handout
- Bozeman explains Lewis structures and VESPR model
- As the pressure increases, the _____ decreases.
Tuesday May 10
AGENDA:
- Chem crash course #12- the ideal gas law- 9:02
- Boyle's Law: P₁V= P₂V₂
- Charles's law: V₁/T₁= V₂/T₂
- Avogadro's Law: V₁/n₁ = V₂/n₂
- P= pressure (in atmospheres) V- volume (in Liters), n= number of moles
- For Boyle's law to hold true, the _____ and the amount of gas must be held constant.
- Boyle's law means that if we know the _____ of a gas at a given pressure, we can predict the new magnitude.
- Charles law: The volume of a given amount of gas (at constant pressure) increases with the ____ of the gas.
- Charles law equation is V₁T₁ = V₂T₂ where the temperature is in the _____ scale
- The volume of gas is directly proportional to the number of _____ as well (if temperature and pressure remain constant). This relationship is called _____ law.
- The Ideal gas law equation is PV=_____ and this is for gasses that are said to behave ideally. This means when pressure is approximately _____ atmosphere or lower and when the temperature is approximately _____ ℃ or higher.
|
zero
temperature nRT |
Kelvin
moles |
pressure
Avogadro's |
1
volume |
Wednesday May 11
AGENDA:
- CHEMISTRY STANDARDIZED TESTING- ACT QUALITY CORE TESTING
- Brainpop: Chemical bonds- van der Waals force. There are actually several kinds of this force, but they all glue substances together in the same basic way: through the uneven spacing of positive and negative charges inside a molecule. London dispersion forces is a form of van der Waals force for non-polar molecules. A good article about electronnegativity is here as well.
- VSEPR and Orbitals
The Orbitron: a gallery of atomic orbitals and molecular orbitals http://winter.group.shef.ac.uk / orbitron /
Friday May 13
AGENDA:
- van der Waals forces- from brainpop: Chemical bonds
- electronegativity- from brainpop: Chemical bonds
- physical properties- from brainpop: Chemical bonds
- Brainpop: ions
- Brainpop: atoms
- Brainpop: property changes
- Brainpop: radioactivity
- Brainpop: temperature
- Brainpop: matter changing states, moles, conservation of mass
- Brainpop: measuring of matter
- Brainpop: Body chemistry
- Brainpop: Water
- neok12.com diagrams for heat and temperature
- neok12.com atoms and matter
- neok12.com: solid, liquid, gas
- neok12.com: nature of matter
- neok12.com: subatomic particles quiz 1
Monday May 16
AGENDA: REVIEWING FOR THE FINAL EXAM
- We are just going to have the final exam and no test before it.
- You have been given a handout for review. I have went over what I WONT cover in it already and I will continue to use this handout this week.
- Chapter 12 will be included on the FINAL. Therefore you should know that ionic and covalent bonding are the extremes of types of bonding and why this is so. You should understand that attractive forces in molecules lie on a continuum with polar-covalent being in between. You should know how they are formed and the nature of their bonds and their relationship to electronegativity.
- You should understand bond polarity and how it is related to molecular polarity
- You should understand factors governing ionic size
- You should learn to write Lewis structures and learn to apply the VSEPR model
- Know about atom structure and orbitals
- Know about coefficients and subscripts and what they are used for
- There will be 25-30 multiple choice questions on the final with I'd say around 25% coming from the most recent chapters. The questions will be conceptual.
- Brainpop: Compounds & mixtures in a compound a chemical change takes place. They don't look anything like their original elements. Mixtures can be separated by physical or mechanical means. Homogenous mixtures are uniform all around. Heterogenous mixtures are where differences are seen between the components. A centrifuge is often used to separate mixtures. So when using a centrifuge, after scientists have spun their mixtures around for awhile, the solid matter lies at the bottom, and the liquid is on the top!
- Brainpop: periodic table of elements- periods go across and describe the number of shells an atom has, the vertical columns are called groups and are numbered 1 to 18. Different colors are organized by their properties. Any element above 92 (uranium) is too unstable and have to be created in a lab. Atomic number = protons
- neok12.com- periodic table : you should know the trends for ionization energy, electronegativity, atomic radii, atomic number. You should know the difference between groups and periods and what each are associated with.
- neok12.com- 5 different reactions- synthesis, single displacement, double displacement, combustion, decomposition
- Test for seniors
Tuesday May 17
AGENDA:
- Brief overview of how atoms are named: Here’s a brief overview of how chemical compounds that consist of only two elements are named.
1. The name of the positive ion always comes before the name of the negative ion.
2. Positively charged ions keep the names of their elements.
3. Negatively charged ions take on the suffix “-ide.”4. Greek numerical prefixes are used when more than one atom of a certain ion is present.
So now, when you hear the term “carbon dioxide,” you can infer that it’s a mixture of a positively charged carbon ion and two negatively charged oxygen ions (the Greek prefix “di-“ means “two”)! Answer: dihydrogen oxide! - Gold, silver and bronze- medals from the olympics 9:42 neok12.com
- 3 different types of heat: radiant energy, conduction, convection
- Brainpop: states of matter- solid, liquid, gas, plasma (electrically charged gas, e.g. lightning, stars, and fluorescent lightbulbs), and colloids (mixtures that contain 2 separate phases of matter e.g. butter, gelatin, and paint), bose-einstein condensateliquids have a fixed volume but not a fixed shape. If we boil water it will become gas. This is done by increasing energy and one way of adding energy is by adding heat to it and the molecules start moving faster and faster.
- Plasma: 99 percent of the visible universe is made of plasma—including every star in the sky—Most of the universe is made up of plasma, a unique, yet common, form of matter. Plasmas are also called ionized gases, and they form when large amounts of heat or other energy cause individual atoms to be stripped of their electrons. When this happens, the atoms become positively charged, and large numbers of free, negatively charged electrons move about within the gas (or other material).
- EXTRA CREDIT - other states of matter: Colloid: A colloid is a distinct state of matter in which very, very small particles—usually less than one micron long—are suspended in some kind of medium, or surrounding substance. There are 3 types and can be translucent (see through) to opaque (can't see through):
- Foams, such as shaving cream, aerosol whipped cream, and ice cream, are air-in-liquid colloids: tiny bubbles of gas trapped in a liquid matrix.
- Emulsions, like mayonnaise and milk, are liquid-in-liquid colloids: particles of one liquid suspended in a medium of another liquid.
- Gels, like Jell-O and toothpaste, are solid-in-liquid colloids: particles of solid matter suspended in a liquid matrix.
- Supersolid- the properties of some solid materials change dramatically when you drop their temperatures until they’re close to absolute zero (0 on the Kelvin temperature scale, or about -273 degrees Celsius). For example, some materials become superconductors; that means they can conduct electricity with little or no resistance. drop the temperature of helium to below 2.176 Kelvin, it becomes superfluid : a liquid that can flow without friction, the resisting force created when two substances rub together. In other words, if you stuck a drop of a superfluid in a can and shook it, the drop would keep flowing around the can until the end of time (as long as you kept its temperature down). And if you cool helium way down to 0.2 K, it becomes a frictionless supersolid—a solid with a superfluid’s frictionless properties!
- -40℃ = -40℉
Wednesday May 18
AGENDA
- Brainpop: acids and bases_
- Acids produce hydrogen ions (H⁺) in water. Bases produce hydroxide ions (OH⁻) in water
- Acids taste sour. Bases taste bitter
- Acids can dissolve metals by taking electrons away from them. Bases do not react with metals.
- Fruit drinks and vinegar are examples that are acidic. Baking soda and soaps are example of things that are basic
Thursday May 19
AGENDA:
- Brainpop: pH scale
- Human body pH is maintained at around 7.4 to 7.5 potential Hydrogen
- Under body level pH of 6.9 this significant drop is called acidosis. Two types. Respiratory and metabolic. Respiratory is with lung disorders such as emphysema and bronchitis. Metabolic is usually trouble with kidneys.
- If your body's pH rises, you can develop alkalosis whose symptoms include light-headedness, nausea, numbness in extremities and muscle spasms.
- blue litmus paper turns red in acidic environments and red turns blue in alkaline environments
- Matter sorter- like tetris but drop objects into 4 different states of matter
Friday May 20
AGENDA:
- van der Waals forces- from brainpop: Chemical bonds
- electronegativity- from brainpop: Chemical bonds
- physical properties- from brainpop: Chemical bonds
- Brainpop: ions
- Brainpop: atoms
- Brainpop: radioactivity
- Brainpop: temperature
- Brainpop: matter changing states, moles, conservation of mass
- Brainpop: measuring of matter
- Brainpop: Body chemistry
- Brainpop: Water- water is known as the universal solvent which means that it dissolves almost anything. Hard water has other metals dissolved in it. Soft water is water lacking in those same minerals. Its the main ingredient of our blood and covers over 70 % of planet's surface. It has polarity. - bottled water shown not to be more beneficial than regular tap water. Water is unique in that when it freezes its volume expands by 9 percent. Water robs fire of the oxygen because of its expansion to water vapor.
- Brainpop: property changes- physical change is change in size, shape or form. Or change in state of matter. chemical change = rusting, tablesalt from the chlorine and sodium gasses (which are toxic individually)d- statue of liberty changed from its original color because of chemical change (oxidation)
Monday May 23
Chapter 12
AGENDA:
- EXAMPLE- HOW TO DRAW LEWIS STRUCTURE AND DETERMINE GEOMETRIC & MOLECULAR SHAPE FOR NITRITE ION_
- geometric and molecular shapes explained
- CHEMISTRY FINAL