2017-2018 FALL SEMESTER
THURSDAY MAY 3
- SOLUTIONS PACKET
- GREATEST DISCOVERIES WITH BILL NYE
- CHAPTER 16 LESSON CHECKS CAN BE WORKED ON
TUESDAY MAY 1
- OBJECTIVES:
- Identify the factors that affect how fast a substance dissolves
- Describe the equilibrium in a saturated solution
- Describe the factors that affect the solubility of a substance
- Identify the factors that affect how fast a substance dissolves
- CHAPTER 16: SOLUTIONS
- Solutions are homogenous mixtures.
- Solubility depends on 3 things: 1) agitation (stirring) 2) temperature 3) particle size of solute
- Solubility is often expressed as grams of solute per 100 grams of solvent or for gases grams of solute per liter.
- know saturated vs supersaturated vs unsaturated
- know miscible vs immiscible
- AS temp goes up a solid or liquid becomes more soluble.
- HENRY'S LAW: In the case of gasses it is the opposite as gasses become less soluble. Its solubility is directly proportional to its pressure of the gas above the liquid. S₁/P₁ = S₂/P₂
- MOLARITY
MONDAY APRIL 30
- CHAPTER 15 TEST (WATER AND ITS PROPERTIES)
- Standardized Review questions as promised
- CK12.ORG Solutions
THURSDAY APRIL 26
- BRAINPOP: FATS
- Carries more energy than any other thing you eat (9 calories/gram) Plutonium is 19.6 million per mole
- made of fatty acids and glycerol
- saturated fats (from most animals) contains cholesterol which can build up in your bloodstream- stay solid at room temp
- unsaturated fats (vegetable oils) considered good- stay liquid at room temp
- trans fat- high level of cholesterol- hydrogenated oil= lots of trans fat
- there is brown fat found in most babies and hibernating animals they have extra mitochondria
- In order of fat content n food from least to most: fat-free = 0.5g ; low fat = 3g; reduced fat = 25%; LIGHT FATY= 50%
- Carries more energy than any other thing you eat (9 calories/gram) Plutonium is 19.6 million per mole
- BRAINPOP: WATER
- Known as universal solvent
- hard water (has Ca, Mg, and other metals dissolved metals in it and causes scum)
- soft water doesn't have much dissolved metals
- covers 70% of earth surface
THURSDAY APRIL 19
- 2 HANDOUTS (ONE ON NUCLEAR RADIATION AND THE OTHER SOLUTIONS)- Start with the nuclear packet as that holds a higher priority the go to the Solution packet. Try to complete as much as you can. They will be a good practice for the test Friday. However, if you are able to finish most of these problems on each packet with correct answers, I will raise one your scores by dropping a low score and giving an "A" instead. We are coming up to the end of the year and that means Finals and so these packets will be great study guide for that as well so hold on to them.
- Chemistry: Greatest discoveries with Bill Nye, Science Documentary- 44 min
- BBC HISTORY: chemistry; a volatile history episode 1 - 1 hour
TUESDAY APRIL 17
- FISSION- the splitting of nuclei into smaller pieces.
- Atomic bomb used neutrons to hit U²³⁵ atom forming U²³⁶ which splits into Kr-91 and Ba-142 + gamma + 3 more neutrons. This is an uncontrolled chain reaction.
- Nuclear power plants use controlled reactions to produce electricity using coolant to absorb the heat produced. The steam drives a turbine which drives a generator that produces electricity. Neutron must be slowed. And it is the water that does this by acting as a moderator. Fuel rods act to absorb the neutrons, decreasing their amount. They are a major cause of nuclear waste. These are made of Pu-239 and U-235
- Produces considerable nuclear waste
- FUSION- When small nuclei combine to make a nucleus of greater mass. In the sun hydrogen (4) combine to become helium + 2 positrons
- Fusion releases much more energy than fission however the temperature is very high to start
- Fission could start fusion, reaching the desired temperature of 40 million ℃. However methods to control and confine this reaction need to be developed. It would produce little radioactive waste (fusion) if controlled
- THE MYSTERY OF MATTER: "OUT OF THIN AIR" (DOCUMENTARY)- 56 MIN- CONTINUING FROM 29:11
- Both chemical and nuclear reactions produce a more stable product
- Pressure, temperature, and catalysts can affect chemical reactions but not nuclear
- Mass is conserved in chemical reactions but not nuclear
- ⁴₂He = alpha particle; º₋₁e = beta particle; ₊₁ºe = positron
- 4 types of radioactive decay: beta emission, electron capture, positron emission, and alpha emission
- Uranium 238 = alpha emission; Uranium 235 = alpha and gamma emission
- Carbon 14 = beta emission; Potassium 40 = beta and gamma emission
- Radium 226 = alpha and gamma emission
- Thorium 234 = Beta and gamma emission
MONDAY APRIL 16
- CRASH COURSE CHEMISTRY #1: THE NUCLEUS
- RELATIVE ATOMIC MASS used to be called atomic weight and is the number of neutrons + protons
- ANTION LEVOSIER- 7 MIN
- The guy that invented chemistry was Antione LeVosier who was put to death. He was called "The Father of Modern Chemistry"
- Due to political upheaval he was put to death.
- He was also a tax collector
- THE MYSTERY OF MATTER: "OUT OF THIN AIR" (DOCUMENTARY)- 56 MIN-
- PHLOGISTON- imaginary element that was used in combustion and it was Lavosier who made precision scales to discover "the conservation of matter" and this made nonsense of phlogiston.
- Lavosier realized that after combustion occurred the product weighed more than the reactant! This was the opposite of what was thought.
- air, water, earth, and fire were all exposed to be made of oxygen.
FRIDAY APRIL 13
- Explained on board alpha particles, beta decay, and gamma radiation, their charges, their mass, and their energy.
THURSDAY APRIL 12
- THE HIROSHIMA AND NAGASAKI FILM THEY DIDN'T WANT US TO SEE- 3:26
- THIS 1946 FILM SHOWS HOW THE ATOMIC BOMB DESTROYED- 12 MIN
- HITLER REACTS TO THE ATOMIC BOMB- 4:50
- 5 DECLASSIFIED NUCLEAR EXPLOSIONS CAUGHT ON FILM
- 10 THINGS YOU DIDN'T KNOW ABOUT HIROSHIMA ATOMIC
- The principle role of the neutrons is to act as a sort of nuclear cement to hold the nucleus together via the strong force.
- This strong force is only over a very small distance
- The more protons there are the more neutrons are needed to hold them together.
- Anything beyond 83 protons (Bismuth), even the addition of extra neutrons cannot stabilize the nucleus
- 3 different types of radioactive decay
- alpha particles - 2 protons and 2 neutrons (nucleus of He atom) and are positive in charged, can be blocked by paper
- beta rays are just streams of electrons and are negatively charged, can be blocked by aluminum
- gamma rays are pure energy, can be blocked by lead and denser material
- U-238 common form of Uranium, U-235 has faster decay rate and used in atomic bomb
- Rutherford did first artificial transmutation of element in 1919 (Nitrogen into Oxygen with Hydrogen left over)
- How many neutrons does the common form of Hydrogen contain? (answer: one)
- Carbon 14 dating works with wood and skeletons less than 50,000 years old. Half life is 5730 years.
- Uranium dating is used for older things. It is based on the lead isotopes Pb-206 and Pb-207 present which is not the common Pb-208
TUESDAY APRIL 10
- 1 atm = 760 mm Hg= 101.3 kPa, .... what pressure, in atmopheres and in mm Hg, does a gas exert at 52.9 kPa?
- What pressure, in mm Hg and atm, does a sample of neon gas exert at 75.0 kPa?
- Demonstrate cotton balls, one soaked with rubbing alcohol and the other water. Which evaporates more quickly? Which has greater vapr pressure at room temperature?
- Although the same temperature, will boiling liquid or the water vapor burn more intensely? (answer: water vapor, higher stored energy)
- What is the strongest material in the world? fullerene nanotubes (made of carbon)
- Why are certain elements radioactive? (unstable nuclei)
- In beta radiation, one of the neutrons in the nucleus of an atom changes into two parts. What are they?
- horizon- hitler's atomic bomb (1992) Enriched uranium quite a bargaining chip in 1944/45 - 50 min
- Uranium 235 could be split by a neutron and cause a chain reaction. Layering it with graphite would make the Uranium 238 more likely to hit another 235 nucleus
- It was perspective in the reasoning that the German's quit atomic bomb research and the americans didn't
- NAGASAKI ATAOMIC BOMB VS HIROSHIMA ATOMIC BOMB- DIFFERENCE BETWEEN NAGASAKI ATOMIC BOMB AND HIROSHIMA
MONDAY APRIL 9
- TEST FRIDAY WILL BE ON WHAT FOLLOWS:
- CHAPTER 25
- Crash courses #27, 28, and 29
- Cathode ray are a ray of electrons
- Liquids with higher vapor pressure will evaporate more easily
- In an open system, there is more evaporation than condesation and eventually all the water will evaporate. In a closed system, the vapor accumulates abo e the liquid, which increases the rate of condensation. Usually the rates of evaporation and condensation will be equal
- Crash course 28
- half life
- 3 types of radioactive decay
- uranium 238 is 98% of uranium decays into thorium 234 and alpha particle (2 protons and 2 neutrons)
- U = He + Th
- beta decay (releases electron)- Th = e + Xe
- gamma decay does not release mass, just releases energy
- Spontaneous fission is when elements split into two on their own but it is very very slow.
- Crash course 29
- The energy that holds the nucleus together is the binding energy and is the "m" in E=mc²
- Anything above element 56 would most likely be involved with fission
- Anything below element of atomic mass 56 (Fe) would most likely be involved with fusion
- control rods help control the nuclear reactions
- Crash courses #27, 28, and 29
- CHAPTER 25
FRIDAY APRIL 6
- CRASH COURSE 33 & 34: DOING SOLIDS
- Amorphous solids melt over a range of temperature and they are not connected in regular ways like the crystalline solids (arranged in orderly ways) o(either ionic or covalent)
- Atomysmal
- #35: a chain in 2d, or a sheet (2d); sigma bonds (not good conductor of electricity, pi bonds better than sigma for conict
THURSDAY APRIL 5
- CHAPTER 13
- air pressure is measured with a barometer- units are 1 atm = 760 mm Hg = 101.3 kPa
- As evaporation takes place the temperature of the liquid decreases, therefore it is a cooling process.
- A measure of the force exerted by a gas above a liquid is called vapor pressure
- when a liquid is heated until particles throughout the liquid have enough energy to evaporate, the liquid boils. The temperature at which the vapour pressure of the liquid is equal to the external pressure on the liquid
- Boiling points decrease at higher altitudes
- Boiling, just as evaporating, is a cooling process,
TUESDAY APRIL 3
- The Strange properties of crystal- 8:50
- The most orderly form of matter in the universe
- All forms of energy will produce a response from the crystal- it will oscillate
- pizeoelectric effect- when you squeeze a crystal it will produce an electric charges or vice versa when charge produce mechanics
- Everything vibrates at a certain frequency
- sugar, salt, street drugs (meth, cocaine), metal, etc... are also crystals
- CRYSTALS AND SCIEND; ENERGIES, FREQUENCIES AND VIBRATIONS- 10:45
- Rochelle salt has greatest degree of pizoelectricity, then tourmaline, then quartz crystal
- quartz crystal has sides and in its natural form has pointed ends
- the thinner the crystal cut the higher the frequency of oscillation
- 396, 417, 528 repairs dna, 639, 741, 852, 963
- CRASH COURSE CHEMISTRY: SILICON
- Silicon is most abundant element found on earth's surface
- form basis of semiconductors
- are responsible for on-off coding responsible for binary coding (transistors)
- SiO₂ IS silicon dioxide
- based on tetrahedral arrangements
- quartz, clays, glass, ceramics (not a crystal) and others
- glass can be made into various shapes (amorphous)
- silicon doping involves putting impurities and made into semiconductors (n-type or p-type)
- when p-type is placed next to the n-type a diode is formed
THURSDAY MARCH 22
- Brainpop Organic chemistry
- Water is the most popular molecule in body
- glucose is body's main source of energy
- collagen and keratin are structural
- Water intoxication is possible
THURSDAY MARCH 22
TUESDAY MARCH 20
- Brainpop: Fats
- Too much saturated fats is bad because it contains cholesterol which stick to the sides of your blood vessels causing plaque. Saturated fats are usually solid.
- Vegetable oil are unsaturated and usually liquid
- Trans fat should generally be avoided
- Fats have the most energy per gram of 9 calories/gram; protein 4 calories/gram; carbohydrates 4 calories/gram
- Brown fat is common in newborns and has ability to generate heat due to its high concentration of mitochondria. Different than the normal yellowish fat.
- Brainpop: Salts
- Consuming lots of potassium- present in fruits and vegetables- can help counteract the negative effects of consuming too much sodium. Can lower blood pressure and prevent kidney problems
- IF you pour salt over a slug it will die due to osmosis
- was used as preservative
MONDAY MARCH 19
- AN EMulsion is two substances that are immiscible together like oil and water
- WHAT YOU NEED TO KNOW ABOUT QUANTUM COMPUTERS AND THE BIRTH OF ARTIFICIAL INTELLIGENCE- 21 MIN
- Instead of using bits the quantum computer uses qubits
- We are receiving a greater amount of energy from other dimensions than we give off
MONDAY MARCH 12
- CRASH COURSES TO COVER (goes along with chapters 15 and 16)
- Water and solutions #7
- bleach is a good oxidizer because it pulls electrons away
- water is an amazing solvent because it is liquid at wide range of temperature, it is very polar and so it is extremely effective at dissolving other polar substances
- water solutions are called aqueous solutions
- dielectric property
- Molarity = mol / L
- Molality = mol / kg
- Acid-Base reactions in solution #8
- 1 of 3 main reactions that happen in solution
- most common base = water; most common acid is water
- anything that donates a proton is acid; anything that accepts a proton is a base
- H⁺ is always H₃O in water solution
- sulfuric acid dissolves limestone
- Redox reactions #10
- The ideal gas law #12
- Ideal gas problems #13
- Real gases #14
- Partial pressures and vapor pressure #15
- Passing gases: Effusion, diffusion and the velocity of a gas #16
- Energy and Chemistry #17
- Enthalpy #18
- Calorimetry #19
- Entropy #20
- Lab techniques and safety #21
- liquids #26
- Solutions #27
- Equilibrium #28
- Equilibrium reactions #29
- pH and pOH #20
- Buffers, the acid rain slayer #31
- Kinetics #32
- Doing solids #33
- Network solids and carbon #34
- Silicon #35
- Electrochemistry #36
- history of atomic chemistry #37
- nuclear chemistry #38
- nuclear chemistry part 2 #39
- hydrocarbon power #40
- alkenes and alkynes #41
- Water and solutions #7
FRIDAY MARCH 9
- TEST OF CHAPTER 12 STOICHIOMEYRY:
- Questions 57, 64, and 65 and, allowing for tim, #60 for count for 25% of the last test
MONDAY MARCH 5
- Brainpop: Metals
- lithium salts used to be present in a number of soft drinks! (It has anti-depressant properties) An early version of Coca Cola called “Lithia Coke” was available at pharmacies. And the drink 7-Up was actually originally called “Bib-Label Lithiated Lemon-Lime Soda!”
- Aluminum used to be considered a precious metal? That’s because even though aluminum is common, the processes for extracting it from its ore used to be very expensive. In fact, Napoleon III of France (pictured) used to serve dinner to his most distinguished guests on aluminum plates; lesser guests had to be content with gold plates!
- lithium salts used to be present in a number of soft drinks! (It has anti-depressant properties) An early version of Coca Cola called “Lithia Coke” was available at pharmacies. And the drink 7-Up was actually originally called “Bib-Label Lithiated Lemon-Lime Soda!”
- Test Friday Stoichiometry including Limiting reagents and percent composition
THURSDAY FEBRUARY MARCH 1
MAKE UP TEST QUESTION:
CONVERSION FACTORS IN COMMON USE FOR CHEMISTRY (UP UNTIL TODAY)
- Rust is produces when iron reacts with oxygen: 4 Fe(s) + 3 O₂ (g) ► 2 Fe₂O₃ (s)
- How many grams of Fe₂O₃ are produced when 12 grams of iron rusts?
- For completeness 100% you need to put your answer in scientific notation with significant figures and correct unit(s).
CONVERSION FACTORS IN COMMON USE FOR CHEMISTRY (UP UNTIL TODAY)
- MOLAR MASS = grams per mole (g/mol) so if you get something in grams you multiply by (1 mole of that substance / #g of that substance (which you get from periodic table)
- AVAGADRO'S NUMBER: you use for the amount of particles of something (e.g. molecules, single elements, compounds) so if you get something in moles (and if you don't you have to get to moles) then multiply by (6.022 x 10²³ molecules or elements or compounds) / (1 mole of those molecules or single elements or compounds). If the question asks you for the number of a specific atom in a compound (elements within a compound or molecule) you would take one step and multiply by (# atoms in the molecule / 1 molecule) e.g. for H in H₂O it would be (2 atoms of H)/ (1 molecule of H₂O
- If you need to find the volume of any gas at STP then you would get to moles and then multiply by (22.L)/(1 mol of the gas)
- MOLE TO MOLE RATIO: You use the coefficients in a balanced equation and multiply by mole2mole ratio with (# moles of substance asked about) / (# moles of given). If you are given a balanced equation such as (6CO2 + 6H2O + light energy = C6H12O6 + 6O2) and you are asked for the amount of a reactant needed or amount of a product obtained (in grams or moles) you would first need to start off with the given amount of something in moles. For example if they asked you for amount of H₂O (in moles) needed to obtain 8 moles of C6H12O16 you would start out with the given: 8 mole C6H12O16 and multiply by mole/mole ratio (6 moles H2O)/(1 mole C6H12O6). There you have your answer in moles. If grams were wanted you would then do a molar mass conversion.
FRIDAY FEBRUARY 23
- Brainpop: The Mole
- A mole of an element equals its atomic mass units weight in grams
- A mole is avagadro's number of anything which is 6.022 x 10²³
- Brainpop: Marie Curie
- a natural process of certain atoms
- substances that give off rays of energy (radiation)
- She noticed that pitchblend gave off more energy the uranium contained within it. These were due to elements #84 and #88, Polonium and radium. Marie Curie discovered them both in 1911.
- She died of a blood disease that can be caused from radioactivity
WEDNESDAY FEBRUARY 14
- Crash course chemistry #6: Stoichiometry: Chemistry for massive creatures
- Stoichiometry is what we use to translate the very small into something very big.
- 1 amu = 1/12 of the mass of a ¹²C atom.
- relative atomic mass of Carbon is 12.01. Used since 1961
- isotopes were discovered
- 1 mole of any element equals its weight in grams
- Crash course chemistry #37
- electrons used to be called corpuscles
- Crash course chemistry #38: Nuclear chemistry, radioactivity
- We can harness nuclear chemistry to produce electricity
- When a nucleus decomposes to form a different nucleus- Radioactivity
- 1/2 life is amount of time it takes for a substance to
- U238 is most common Uranium isotope and it, by aldspha decay (2neutrons=2protons), turns to Thorium234.
- Beta decay is an electron eg (Thorium234
- gamma decay releases just energy so it has no mass (can penetrate your skin and even organs, alter dna)
- spontaneous fissium (Californium is the only one that does this at an observable rate)
- CHEMISTRY- Greatest discoveries with Bill Nye, Science Documentary- 44:04
- Avogadro realized that gasses were made out of molecules and not just elements
- In the old days they used to call "gasses" airs.
- Uranium breaks down into Radium and Plutonium
- THE NOBEL PRIZE EXPLAINED: 13 INTERESTING FACTS
- physics, medicine, physiology & medicine, literature, and peace
- QUESTIONS FOR ACADEMIC BOWL DAY 2
- The number 6.022 x 10²³ is called a.... a) Pi b) Phi c) a mole d) parsec
- What is the lightest element?
- What does a light year measure?
MONDAY FEBRUARY 12
- How many carbon atoms are in 93.3 grams of propane (C₃H₈ )?
- Given: 93.3 grams propane
- What units will my answer be in? Just the number of carbon atoms, no units.
- first conversion factor I will use molar mass of propane is (1mole propane/44grams propane)
- 2nd conversion factor is (6.02 x 10²³ molecules of propane)/(1 mole propane)
- 3rd conversion factor is (3 Carbon atoms/ 1 mole propane)
- 93.3g pro (1 mole pro/44g pro) (6.02 x 10²³ molecules pro/1mol pro) (3 Carbon atoms / 1molecule pro)
- answer is 38.2 x 10²³ atoms of carbon
WEDNESDAY FEBRUARY 7
- piezo popper kit demo on youtube
TUESDAY FEBRUARY 6
- nitinol wire/shape memory alloy- how to use it- 3:11
MONDAY FEBRUARY 5
- WORKSHEET HANDED OUT DUE BY FRIDAY (THE DAY OF THE TEST)
- Went over a couple percent composition problemso
WEDNESDAY JANUARY 31
- BBC CHEMISTRY A VOLATILE HISTORY EPISODE 3
- Rutherford realized that nuclear reactions changed one element into another
- Enrico Fermi then realized that if you shoot neutrons at the nucleus instead of just protons this might help break down the nucleus in that neutrons would not be repelled as alpha particles would (2 protons and 2 neutrons).
- He also noticed that if you shoot enough alpha particles then the mass would start becoming to big and one of the neutrons might change into a proton which would change the entire element into another element the next higher up atomic number
TUESDAY JANUARY 30
- Only 92 elements makes up the whole universe. Element 92 is iron.
- BBC CHEMISTRY A VOLATILE HISTORY EPISODE 2
- Iron is the earth's most abundant element IN OPP
MONDAY JANUARY 29
- BBC CHEMISTRY- A VOLATILE HISTORY EPISOD2 - PERIOD 2
- OCTOPUS AND SNAIL AND SPIDERS HAVE BLUE BLOOD WHICH IS COPPER BASED.
- assigned problems 50-61 as homework, did 52(a) on board
FRIDAY JANUARY 26
- BBC CHEMISTRY- A volatile History- Discovering the elements episode 1- 60 min- left off at 46:38
- Everything is made up of 92 elements
- An element is a substance that can't be broken down
- Alchemists goal was to turn metal into gold. Challenged idea that everything was made from earth, fire, wind, and water.
- Paraselses (1600s) saw 3 elements of salt, sulfur and mercury (known then as quicksilver) as making up everything,
- Liquid mercury is 14x heavier (steel floats on it) than water and mercury is the only metal that is liquid at room temperature
- people suffered from mercury madness
- Man (Brant) - 1600S- looking for gold in urine ended up finding Phospshate.
- phosphorous when used as light source is cool to the touh
- robert boyle by accident found that striking phosphate caused fire (lead to matches)
- Henry Kavendish was rich, painfully shy, and had autism. He was first to discover gas (instead of just f.
- He collected the (inflammable air) from the bubbles being made off zinc and found hydrogen. He thought it was flogistant so his name was not used in the nammer of it.
- He also found that water was not an element but rather a compoud
- By 1700s there was common air, fixed air and ???? ROUND 32 IN
- PRIESTLY was the man of airs and discovered carbon dioxide then oxygen.
- oxygen at -183℃ turns to pale blue liquid. He did not realize he had discovered an element but his information lead to La
- Antoione La Voiselle weighed substances very accurately. When tin was heated it weighed more. Therefore he realized that a substance takes in oxygen and then releases it as a product of combustions. This made flogestin known as non-existant. He was the first to define an element: that which could not be broken down (gases, metals, nonmetals, earth)
WEDNESDAY JANUARY 24
- The mole
- What is the mass of 5.0 x 10²¹ molecules of water?
TUESDAY JANUARY 23
- Kahoot- Basic Chemistry 49 questions
- Still just sections 12.1 and 12.2 going over and over
TUESDAY JANUARY 15
- Handout of molar conversion probems
FRIDAY JANUARY 11
- ck12.org molar mass conversions
- assigned #53, 57, 58, 61 (A and B answers for each one) what is not done will be homework due Tuesday
WEDNESDAY JANUARY 10
- COMPLETED CK12.ORG MOLES
- IN CLASS ASSIGNMENT: #13 AND #14 FROM LESSON CHECK 1 DONE EXACTLY THE WAY I DO THE PROBLEMS IN CLASS
TUESDAY JANUARY 9
- Practiced problems
- Went through 10.1 and 10.2
- 3 conversion factors so far:
- 1 mole = 6.02 x 10^23 molecules or atoms
- molar mass (grams/mol)
- At STP for gasses 1 mol of any goas = 22.4 L
- HOMEWORK LESSON CHECK 10.1
SEMESTER TWO
MONDAY JANUARY 8
- STARTING CHAPTER 10: STOICHIOMETRY
- Brainpop: Mole = 6.02 x 10^23
- Mole day is October 23 starting and ending at 6^23
- Crash course chemistry #6: Stoichiometry-
- Conversion factors:
- 1 mole = 6.02 x 10^23 molecules, compounds or atoms of something
- grams per mole ... eg.... 1 mol C = 12 grams C
MONDAY DECEMBER 18
- ck-12.org
- Scientific notation
- States of Matter: Solids, liquids and gasses
- homogenous and heterogenous mixtures
- How to recognize chemical reactions (light, heat, bubbles, precipitate, change of color)
- know units for density
- 1 mL = 1 cm³= 1 cc
- Atomic number:
- Bohr's atomic model: To go from low to high orbital electrons need to gain energy; high to low releases energy in form of light (photon)
- Valence
- periodic trends
- Molecular formula
- formjlas for binary ionic compounds
- Binary molecular compounds: naming
- Naming acids
- ionic bonds
- Lewis electron dot structure
- basic reaction types
MONDAY DECEMBER 11
- Went over three rules for naming acids
FRIDAY DECEMBER 8
- LABORATORY: Using different acqueous indicators (Phenolpthalein, bromoblue, and orange) and pH testing
WEDNESDAY DECEMBER 6
- GENERAL RULES FOR SOLUBILITY OF IONIC COMPOUNDS (SALTS) IN WATER AT 25℃
- Most nitrate salts are soluble
- Most salts of Na⁺,K⁺, and NH₄⁺ are solubleS
- Most chloride salts are soluble. Notable exceptions are AgCl, PbCl₂ and Hg₂Cl₂
- Most sulfate salts are soluble. Notable exceptions are BaSO₄, PbSO₄, and CaSO₄
- Most hydroxide compounds are only slightlt soluble. The important exceptions are NaOH and KOH. Ba(OH)₂ and CA(OH)₂ are only moderately soluble
- Most sulfide, carbonate, and phosphate salts are only slightly soluble.
- *** The terms insoluble and slightly soluble reallhy mean the same thing. such a tiny amount dissolves that it is not possibe to detect it with the naked eye.
- CRASH course chemistry #9: Precipitation reactions
- The latin word for silver was argentum
- Silver has anti-disease properties
- Silver nitrate and silver??? were both used as antiseptics during wars of the olden days. Some also use colloidal silver.
- Crash course chemistry #11: How to speak Chemistrian
|
|
TUESDAY DECEMBER 5
- Brainpop: Acids and Bases
- Rainwater has pH about 5 or 6 with pollutants like sulfur dioxide and various nitrogen oxides. They combine with water to make sulfuric and nitric acid.
- Acids release H+ in water forming H₃O⁺. Acids are sour. Carbonic acids eat away and causes stalagtites
- Distilled water is 7 pH which is neutral
- Bases feel slippery and taste bitter. They don't react with metals like acids do so they won't destroy them.
- To combat heartburn compounds of magnesium, aluminum, or sodium bicarbonate is taken to neutralize the pH.
- Vinegar: acetic
- Spinach: oxalic acid
- soft drinks: carbonic acid
MONDAY DECEMBER 4
- Played polyatomic ions, molecule or ionic bond, and which two make a precipitate
- Explained how acids and bases are named (H+ and OH-)
- the "ate" becomes "ic" followed by acid
- e.g. sulfate becomes sulfuric acid
- the "ite" becomes "ous" followed by acid
- e.g. sulfite becomes sulfurous acid
- the "ide" becomes "hydro" followed by "ic" followed by acid
- e.g. chloride becomes hydrochloric acid
- the "ate" becomes "ic" followed by acid
- Ion charges (including transitional and polyatomic ions)
WEDNESDAY NOVEMBER 29
TUESDAY NOVEMBER 28
- sulfate and sulfite differ by one oxygen (Sulfite has one less oxygen). This is same for phosphate/phosphite , nitrate/nitrite, etc....
- the charge of a transition metal can be found by "ous" or "ic". For example Tin can be either a 2+ or a 4+ charge. So Stann"ous" chloride would have the lower charge and stann"ic" would have the higher charge.
- The same can be applied to acids as we will learn tomorrow ("ous" vs "ic)aq
MONDAY NOVEMBER 27
Map of Chemistry
- Polyatomic ions to know: nitrate (NO₃⁻) , cyanide, hydroxide, chlorate, sulfate, carbonate, chromate, ammonium, phosphate, mercurous, mercuric, peroxide
MONDAY NOVEMBER 13
- CHAPTER 9 "CHEMICAL NAMES AND FORMULAS"
- Transition elements have more than one ion charge state.
- (I & II) Cu and Hg₂²⁺ (I), Hg²⁺ (II)
- (II & III) Fe, Mn and Cr
- (II & IV) Pb and Sn
- classical names used instead of roman numerals e.g. ferrous (+1 charge) and ferric (+2 charge)
THURSDAY NOVEMBER 9
- Antioxidants and Free radicals- 5 min
- bad free radicals capture electrons from your cells
- antioxidants stabilize free radicals by giving electrons to bad free radicals so they don't need to steal them from you
- free radicals enter through sun exposre cigarette smoke pollutio, pesticides, alohol, fried food, water and medicine
- glutathione is "master antioxidant" in whey protei non-pasteurized ilk, raw fruits and vegeta les, vitamin E and C, beta carotene
- Review 8.4
- Intermolecular and intramolecular bonds- 2:27
- Intramolecular bonds are covalent and ionic and metallic bonds. They are forces between atoms in a molecule
- Intermolecular bonds are bonds betweeds
- Intermolecular forces and boiling points- 11 min
- ion-ion
- ion-dipole
- dipole-dipole (strongest one is hydrogen bonds F, O and N)
- Van der Waals and London dispersion reactions are the same thing (anything can have this force) momentary dipole. They are induced. Halogen diatomic molecules attract each other mianly by means of dispersion forces.
- Boiling points depend on strength of intermolecular forces: State which molecule in each pair has higher boiling point and why
- HF vs HI
- O2 vs CH3Cl
- H2O vs NaCl
- He vs Ar
- TEST CHAPTER 8
TUESDAY NOVEMBER 7
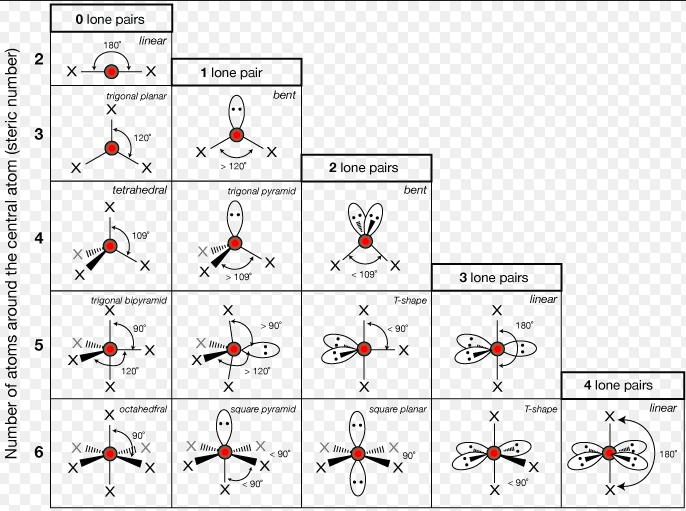
- Drawing the shapes of molecules- 5 min
- Sonic geometry: the language of frequency and form- 32:11
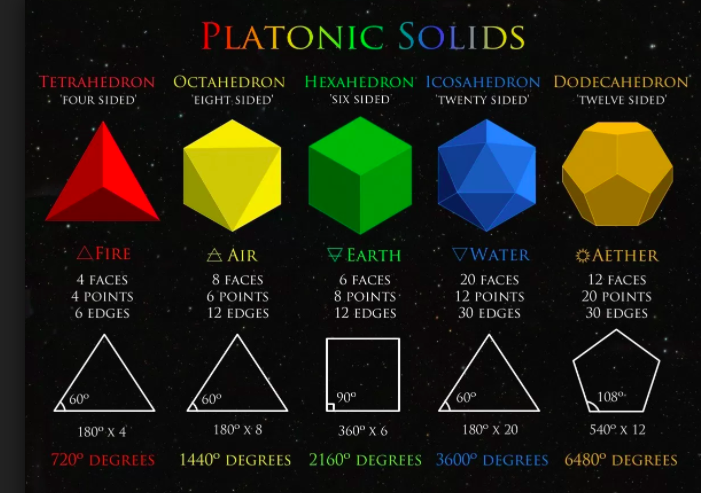
- The elements: Classical elements and platonic solids- 2:30
- The platonic solids: There can only be (phi)ve- (2D EQUILATERAL TRIANGLE FACE) TETRAHEDRON, OCTAHEDRON, ICOSAHEDRON, (2D SQUARE) CUBE, DODECAHEDRON (EUCLID SHOWED 5)
- 3 FROM TRIANGLE, 1 FROM SQUARE, 1 FROM PENTAGON
- TETRAHEDRON FIRE, OCTAHEDRON AIR, ICOSAHEDRON WATER, CUBE EARTH, DODECAHEDRON UNIVERSE ITSELF OR AETHER
- 3 properties: all faces are regular polygons, are exactly the same, and all corners are the same
- ALL five platonic solids have been used as dice
- The octahedron is the dual of the cube (appears in the middle of the cube when the points of the cube are joined. The cube has 8 vertices and 6 faces and the octahedron has the opposite
- the dodecahedron is the dual of the icosahedrons and vice versa. This is called duality. The icosahedron has 20 faces and 12 vertices.
- the tetrahedron is a dual of another tetrahedron
MONDAY NOVEMBER 6
- Quizlet Molecular geometry
- Molecular shape and polarity
- The following shapes may be nonpolar: linear, trigonal planar, tetrahedral, and trigonal bypyramidal, octahedral, square planar
- Molecular structures
- The tetrahedron, cube, and octahedron all occur naturally in crystal structures.
FRIDAY NOVEMBER 3
THURSDAY NOVEMBER 2
- video- Ionic, covalent, and metallic bonds- from ck12.orgl
- Bozeman science Metallic bonding- 5:54
- Color of metals by dissolving metals in soslution. sodium is a yellow orange color; strontium is crimson red; Barium is apple green; copper is blueish greens
- Rainbow flame; coloured fire experiment: NaCl yellow; liCl (red); StCl2; H3 BO3; cuso4; bacl2 greens; pbno32 kcl (turns more purple at end ch3OH is fuel
- Lithium burns red; methanol burns blue all by itself; Lithium (deep red); Calcium (orange); Na is yellow ; Boric acid has copper and will burn green, KCl burns purple n
- Clear quartz: The master crystal
TUESDAY OCTOBER 31
- Intra vs Inter molecular bonds
- (Intramolecular bonds are ones between each atom in the molecule)
- (Intermolecular bonds are the ones between each molecule)
- London dispersion forces occur in all nonpolar substances
- Crash course chemistry # 24: Bonding models and lewis structures
- The power of quartz crystal - 10 min
how to find and use quartz for survical- 7 min
MONDAY OCTOBER 30
- BOZEMAN DRAWING LEWIS DOT DIAGRAMS
- They tell a lot about valence electrons and structure of atom.
- was a nobel prize contestant 35 times but didn't once win.
- Determine the total number of valence electrons
- Arrange the atoms' symbols (Carbon atoms are in center). Usually it is most electronegative in center
- Draw lines connecting the atoms (one line = 2 electrons)
- Compare the number of valence electrons in structure with number derived in #1
- If not balanced then start double bonding
FRIDAY OCTOBER 27
- handout ionic compounds and also of covalent compounds.
THURSDAY OCTOBER 27
- CHAPTER 7 TEST- STANDARDIZED
- LABORATORY-
- Tried some of the water demos
THURSDAY OCTOBER 26
- Crash course chemistry #7: Water and solutions for dirty laundry- 13:33
- Crash course chemistry # 22: Atomic hookups: types of chemical bonds- 9:45
- LABORATORY
- DOES HOT WATER RISE
MONDAY OCTOBER 23
FRIDAY OCTOBER 20
- Chemistry of energy storage materials
- How would you say the following compounds:
- MgCl₂
- Al₂S₃
- CaI₂
- BaCl₂
- Li₂O
- Metals are good conductors while ionic compounds re only good conductors when they are molten or dissolved in water.
- Metals will flatten but not break because the cations and electrons are mobile and the ionic substance will shatter. It is these mobile valence electrons that explain a metal's ductility and malleability
THURSDAY OCTOBER 19
- Write the chemical formulas for the compounds formed when the following elements combine:
- magnesium and chlorine
- aluminum and sulfur
- calcium and iodine
- barium and chlorine
- lithium and oxygen
- Brainpop: Crystals
- liquid crystal is another phase of matter
- Crystals- bell laboratories- 24 min
- Ionic bonds form when one or more _____ are transferred between atoms. The atoms that lose electrons become _____ and the atoms that lose electrons become _____ and the atoms that gain electrons become _____. Both these ions have an electron configuration with a _____.
- Kahoot: Ionic bonds (extra credit for winner)
TUESDAY OCTOBER 17
- WORK on homework section reviews and packet
MONDAY OCTOBER 16
- PRACTICE QUESTIONS IN BOOK
FRIDAY OCTOBER 13
- PRACTICES NAMING IONIC COMPOUNDS
THURSDAY OCTOBER 12
- GROWING CRYSTALS LAB
TUESDAY OCTOBER 10
- TEST CHAPTER 6 "PERIODIC TABLE"
- Crash course Chemistry #22 "Atomic hook-ups: Types of chemical bonds"
- What are ionic bonds? 2:54
MONDAY OCTOBER 9
- REVIEW FOR TEST TOMORROW
FRIDAY OCTOBER 6
- PERIOD TRENDS
- ELECTRONEGATIVITY- Fluorine has the highest
- ATOMIC RADIUS- He is the smalest and Fr is the biggest
- IONIZATION ENERGY: amount of energy it takes to remove an electron- He has the largest, Fr is the least
- ELECTRON AFFINITY: the energy released when electron is added to an element- how much element wants to gain electron- Fluorine is the highest
THURSDAY OCTOBER 5
- 5 CRAZY TRICKS WITH WATER
- OOBLECK (PART SOLID PART LIQUID)
- How a Russian guy's dream changed the science world- 8:08
- History of the periodic table animation- 6;38
- 1829- Doberiegn's triads
- every eighth element had similar properties "Newman Octaves- only up to Calcium
- Mendeleev used atomic mass in classifying in increasing order with 63 elements.
- There are a total of 7 periods
- There are 8 groups
- demerits: no fixed position could be given to Hydrogen, sometimes atomic mass lighter than one had to be switched to be consistent with the table
- 1913- Henry Moseley discovered that the atomic number was the most important number and not the mass.
TUESDAY OCTOBER 3
- CK12.org Practice assessment questions about periodic table
MONDAY OCTOBER 2
- BRAINPOP: MARIE CURIE
- She discovered Plutonium and Radium
- She died of cancer
- CK12.ORG
FRIDAY SEPTEMBER 22
- Add on the following elements (in addition to the first 4 rows): Rb, Sr, Cs, Ba, Pd, Ag, Cd, Sn, Sb, I, Xe, Pt, Au, Hg, Pb, U
- TEST CHAPTER 5- ELECTRON CONFIGURATIONS, ORBITALS, QUANTA, AND WAVES
- CRASH COURSE CHEMISTRY #4- THE PERIODIC TABLE
- In the 1860s about 60 elements were known
THURSDAY SEPTEMBER 21
- Brainpop: Electromagnetic spectrum
- The higher the energy the more things the radiation can pass through.
- electrons changing energy levels is what produces electromagnetic radiation
- if there was no ozone layer the rates of skin cancer would skyrocket
- microwaves emit waves with a wavelength of about 12.24cm and are easily absorbed by molecules of sugar and water. So water in food is penetrated and they begin to vibrate rapidly which causes them to generate heat.
- Microwaves are much faster than regular conventional oven because they cook from the inside out rather than the opposite.
- Black lights emanates waves of ultraviolet light. Laundry detergents contain chemicals called phosphors which absorb photons of ultraviolet light and then emit photons of visible flight. They are put in to make white clothes look bright and clean in sunlight. Phosphors are also put into fluorescent fabrics and paints. Modern paints contain phosphors that weren't used before the 20th century. So back lights can detect forgeries.
- Unite states currency contains an invisible fluorescent strip that only shows up under black lights. Crime investigators also use black lights.
- Prior to satellite communications "world band" shortwave radio (between AM and FM) was used for global communication. Ships and planes use shortwave communication as do communities of amateur radio operators known as HAMS.
- Supernovas (exploding stars) and massive black holes give off gamma rays as does radiation therapy and decaying radioactive materials.
- Brainpop: Waves
- Waves carry energy
- Brainpop: Light
- Running electricity throught a solid produces light like in a light bulb
- acts like a wave sometimes and a particle other times (photons).
- 300 million meters in one second. (3.0 x 10⁸ m/s)
- Its speed changes as it travels through different things
- Aurora borealis and Aurora australis" : flashing green lights in sky by electrons going from higher to lower energy
- Bioluinescence is producing light via chemical ractions in bodies. Firefly have light emitting organ in ower abdomen which produce light when lucierase interacts with oxygen and another chemical called lucoiferin
TUESDAY SEPTEMBER 19
- electron configuration practice
- went over third section of chapter (electromagnetic radiation and waves)
MONDAY SEPTEMBER 18
- What does an atom REALLY look like? 8:43
- Crash course chemistry #5: The electron
- Make up test
FRIDAY SEPTEMBER 15
- ELEMENT QUIZ RETRY (HIGHER OF TWO ELEMENT QUIZZES WILL BE SCORED)
- TEST CHAPTER 4
- DEMONSTRATION - PRESSURE CRUSHING CAN
- find video on baking bread making yeast
THURSDAY SEPTEMBER 14
- NATIONAL HONOR SOCIETY SCHOOL VIEWING
- PRACTICED FILLING OUT TABLES: ELEMENT- ATOMIC #- MASS #- #PROTONS- #NUETRONS- #ELECTRONS
- ELEMENT QUIZ
TUESDAY SEPTEMBER 12
- Cathode Ray Tube experiment- it discovered the electron
- Rutherford's gold foil experiment-
- 5 most dangerous elements -10 min
- Mercury, Arsenic, Cesium (radioactive), Polonium, Plutonium
MONDAY SEPTEMBER 11
- FUN FACTS ABOUT ELEMENTS 1 -20
- HYDROGEN- lightest element
- lithium- lightest metal
- fluorine- most chemically reactive
- Mg- used in flash photography
- silicon- used to make glass
- chlorine- corrodes almost everything it touches
- 13 most fascinating elements
- Carbon- makes diamond and graphite
- Chlorine- cl gas used in warfare
- Bromine is liquid at room temperature (can cause chemical burnw)
- arsenic (causes heart disease and thickening of skin)
- gallium (low melting point) cookie mold
- mercury (known as quicksilver) used in thermometers
- cesium (also forms liquid near room temperature
- Tanner suggested baking bread
- 10 mind blowing experiments compilations #7
- blowing cornstarch into a fire makes it a fireball
- copper wire made to twirl on double
- MOST AMAZING EXPERIMENTS YOU CAN USE AT HOME
- Make an electromagnet
- mentos and diet coke mix makes explosion
- oobleck (cornstarch and water with food coloring 2 to 1 ratio corn starch to water) use 40, 50 or 63 yz
- tea bag rocket
FRIDAY SEPTEMBER 8
- HANDOUT POCKET- DUE DAY OF TEST
THURSDAY SEPTEMBER 7
- 118 amazing facts about the 118 elements part 2- 11:25
- Went over worksheet handed out last week (how to convert from cm to cm³)
- DENSITY LAB (must get another scale) and start again as even my personal scale had become inoperable.
TUESDAY SEPTEMBER 5
- Bill Nye the Science guy- atoms- 23 minutes
- Crash course chemistry #1- The Nucleus- 10 minutes
- 118 amazing facts about the 118 elements part 1- 10:45
- For the remainder of class they can start reading the very next chapter (CHAPTER FIVE) in the text.
FRIDAY SEPTEMBER 1
- TEST #2
THURSDAY AUGUST 31
- Lab: Pressure- failed- cite reasons why
TUESDAY AUGUST 29
- INTRODUCTION TO THE ATOM: THE CHEMISTRY OF LIFE
- ORBITALS: ELECTRON STRUCTURE OF ATOMS
- There is a dimensional analysis, sig fig, scientific numbers packet that will help you check what you know.
- Try the following problems:
- The population of California is 3.7 x 10⁷ people. The population of Colorado is 5.0 x 10⁶ people. What is the difference in size between these two populations?
- The density of a substance is 3.4 x 10³ g/mL. Mixing it with another substance increases the density by 1.5 x 10¹ times. What is the density of the mixture?
- A contractor uses 16 tiles to cover a bathroom floor that has an area of 2.4283 m². What is the area of each tile? Give the answer to the correct number of significant figures.
MONDAY AUGUST 28
- PRACTICE QUESTIONS
FRIDAY AUGUST 25
- Any conversion factor such as (2.54cm = 1inch) should not be used when considering significant figures.
- Density (mass/volume) example (grams per Liter) or (grams per cm³) since 1mL = 1cm³ = 1cc
- Weight changes depending on where something is while mass stays the same everywhere.
- The coldest anything can be is absolute zero Kelvins
- Kelvins (K) and Celcius (℃) have the same scale where a change of 1 K = a change of 1℃ . However one must subtract 273 from the amount of Kelvins to get the same temperature in celcius or add 273 to anything celcius to get the same temperature in kelvins
THURSDAY AUGUST 24
- Significant figures (rule for addition/subtraction and multiplication/division)
- Scientific notation (in the form of #.## x (multiplied by) 10^x) which is basically writing very large or very small numbers with a lot of zeros into a more recognizable number. Practice problems 12, 13 and 18
WEDNESDAY AUGUST 23
- TEST ON CHAPTER 2
- CRASH COURSE CHEMISTRY #3 (conservation of mass and Lavoisier's execution)
TUESDAY AUGUST 22
- Went through practice problems, reviewed for tomorrow's test.
MONDAY AUGUST 21
- A diatomic molecule is a stable group of two atoms connected by chemical bonds. Diatomic molecules that are comprised of the same element are called diatomic elements. There are five diatomic elements that exist as gases at standard temperature and pressure: hydrogen, nitrogen, oxygen, fluorine and chlorine. Additionally, at elevated temperatures, bromine and iodine also exist as diatomic elements.
- A good mnemonic to use for remembering the seven diatomic elements is "Have No Fear Of Ice Cold Beer." The first letters of each word corresponds to the atomic symbols of the seven elements: H for hydrogen, N for Nitrogen, F for fluorine, O for oxygen, I for iodine, Cl for chlorine and Br for bromine.
FRIDAY AUGUST 18
- CHAPTER 2 LESSON CHECKS
- TEST SCHEDULED FOR TUESDAY, REVIEW MONDAY
THURSDAY AUGUST 17
- BRAINPOP PROPERTY CHANGES www.brainpop.com/science/matterandchemistry/propertychanges/
- BRAINPOP: SALTS
- They break up in water and conduct electricity. The body cant store salt so we get it by the food we eat.
- It was a popular food preservative. It stopped bacterial growth.
- Was used for currency exchange
- It melts ice on the roads and acts to lower the freezing temperature
WEDNESDAY AUGUST 16
- When a solid material seems to disappear in a liquid, the mixture is called a solution. Solutions are always clear. You can see through solutions. A solution can be colored, like iced tea or lemonade, but it must be clear. Do you think chocolate milk is a solution? Do you think NaCl is a solution?
TUESDAY AUGUST 15
- continue video from 30 min
- Professor Dave General Chemistry: Introduction to chemistry #1 [email protected]
- Professor Dave #2: Types of matter: elements, compounds and mixtures
- Brainpop: Compounds & mixtures;
- Dimensional analysis: page 19 sample problem 11
- READ CHAPTER 2.1 and do 2.1 Lesson check
MONDAY AUGUST 14 FIRST DAY OF SCHOOL
- Hand out texts
- CHEMISTRY- Greatest discoveries with Bill Nye- 44 min finished at 30 min
EXTRA: CHAPTER 16
QUESTION FILL INS CHAPTER 16 "SOLUTIONS"
- A mixture is a _____ solution
- 3 ways to make a substance dissolve quicker
- S1/P1 = S2/P2 What law is this?
- M1/V1 = M2/V This equation is used for making _____
- Colligative properties do not depend on the type but rather the _____ of particles.
- The 3 colligative properties of solutions are _____ lowering, _____ depression, and _____ elevation
- _____ is a solution used in your car to make colligative property changes.
- As the number of particles in a solution increases, the vapor pressure _____, the boiling point _____, and the freezing point _____.
- Which produces a greater change in colligative properties when added to a solvent- a mole of an ionic solid or a mole of a molecular solid?
|
|
|